Never Fall Behind with an Automatic Deblister Machine
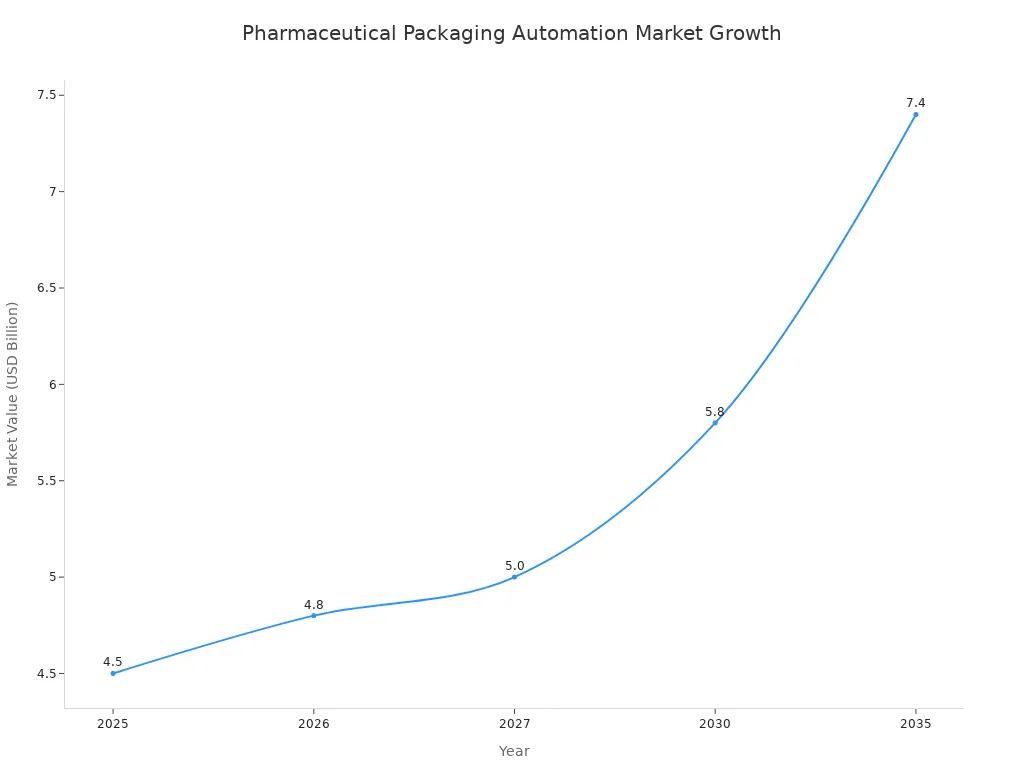
Investing in an automatic deblister machine by 2026 is a critical move to secure a company's future operational viability. The pharmaceutical industry's rapid move toward automation makes this technology a necessity. It helps control costs, ensure compliance, and maintain a competitive edge.
The primary reason to invest is to protect the bottom line. Companies maximize drug recovery, reduce labor expenses, and eliminate compliance risks. Understanding tablet deblistering benefits shows why this pharmaceutical deblistering equipment is essential. A Deblister Machine is no longer an option.
Key Takeaways
- Automatic deblister machines save money. They cut labor costs and stop product waste. This helps companies earn more.
- These machines make workplaces safer. They prevent injuries from repeated hand movements. They also help follow important safety rules.
- Automation helps businesses grow. It makes production faster. It also lets companies use new types of packaging.
- Choosing the right machine is important. It must work with your products. It also needs to be safe and easy to clean.
Drive Down Operational Costs and Maximize ROI
Adopting automation is a direct strategy for improving a company's financial health. An automatic Deblister Machine provides a clear path to a stronger return on investment (ROI) by tackling major operational expenses head-on. Companies see significant financial benefits by reducing manual labor, eliminating product loss, and preventing costly errors.
Slash Labor Costs & Reallocate Talent
Manual deblistering is a time-consuming and repetitive task. It requires significant human resources that could be used for more valuable activities. Automation fundamentally changes this dynamic. It allows companies to reassign skilled technicians from tedious manual work to roles that require critical thinking and expertise. This shift has several key advantages:
- Reduced Overtime: Automation handles high-volume tasks consistently, decreasing the need for overtime pay during peak production periods.
- Optimized Staffing: Fewer operators are needed for the deblistering process, allowing for leaner, more efficient team structures.
- Increased Employee Value: Staff can focus on quality control, machine maintenance, and process improvement, contributing more directly to innovation and growth.
This strategic reallocation of talent not only cuts direct labor expenses but also boosts overall team productivity and morale.
Minimize Product Waste & Recover Valuable Medication
Every lost tablet or capsule represents a direct financial loss. Manual deblistering methods are often imprecise, leading to damaged products that must be discarded. High-value medications, such as those used in oncology or clinical trials, make this waste especially costly.
An automated system uses precision mechanics to gently and effectively push products from their packaging. This process achieves recovery rates of nearly 100%. It ensures that valuable medication is saved and can be repackaged or used, protecting revenue that would otherwise be lost.
By maximizing the recovery of every single dose, a Deblister Machine turns potential waste into tangible assets, directly improving the bottom line.
Quantify Savings from Reduced Manual Errors
Manual processes are prone to human error. In a manufacturing environment, mistakes like incorrect counts or component mix-ups directly harm a business's financial health. These inventory errors lead to higher expenses and documented financial losses. All inventory classes are critical assets. Their mismanagement due to manual mistakes directly impacts a company's balance sheet and ability to generate revenue.
Even minor manual errors in tracking and processing can accumulate into substantial financial damage, potentially reaching thousands or even millions of dollars annually. These losses come from issues like misclassified batches or inaccurate inventory counts, which hurt profitability and slow down growth. Quantifying these potential losses shows the clear need for proactive error management. Automation provides this solution by ensuring accuracy, minimizing financial risk, and protecting a company's reputation.
Enhance Safety, Compliance, and Data Integrity for 2026
Beyond cost savings, automation strengthens a company's foundational pillars of safety and regulatory adherence. A modern Deblister Machine is an essential tool for protecting employees, satisfying stringent government oversight, and securing critical operational data for the future.
Eliminate Repetitive Strain Injuries (RSIs)
Manual deblistering requires constant, repetitive hand and wrist motions. These actions frequently lead to painful and costly repetitive strain injuries (RSIs) for operators. Automating this process removes the human element from the strenuous task. This shift directly protects employee health and well-being. Companies reduce their risk of workplace injury claims and create a safer operational environment for their teams.
Ensure GMP and FDA Compliance with Precision
Meeting regulatory standards is non-negotiable in the pharmaceutical industry. Automation provides the precision needed to adhere to Current Good Manufacturing Practices (cGMP) and FDA rules. Machines designed for compliance prevent cross-contamination and maintain product integrity. Key regulations addressed include:
- 21 CFR Part 11: Governs electronic records and signatures.
- 21 CFR Part 211: Outlines cGMP for finished pharmaceuticals.
These systems are built with materials like SS304 and SS316 stainless steel. Their smooth, easy-to-clean surfaces and validated cleaning procedures ensure hygienic operation, a core requirement for cGMP environments.
Tip: Regular calibration of machine settings and documented cleaning logs are critical practices that support consistent performance and simplify regulatory audits.
Strengthen Audit Trails with Automated Data Logging
Regulators like the FDA and EMA require complete, unalterable records for all manufacturing processes. Manual logs are vulnerable to error and falsification. Automated systems solve this by creating secure, time-stamped audit trails for every action. The system automatically captures data on batches, operator IDs, and operational times. This creates a reliable chain of custody. These secure electronic records cannot be edited, ensuring the data integrity required by 21 CFR Part 11. This automated data logging provides the verifiable proof of compliance needed during inspections and protects a company’s operational history.
Future-Proof Operations Against Market Demands
Staying competitive means preparing for tomorrow's challenges today. Investing in automation allows companies to build resilient operations that can scale with market demands, meet sustainability targets, and handle future packaging innovations.
Increase Throughput for Production & Clinical Trials
The demand for pharmaceuticals is constantly growing. Companies must increase their production speed to keep up. Automation is the key to unlocking higher productivity and efficiency. An automated system significantly speeds up the deblistering process for both large-scale manufacturing and time-sensitive clinical trials. This increased speed offers several advantages:
- It boosts overall productivity and accuracy.
- It reduces labor costs associated with manual work.
- It enables the efficient preparation of patient-specific medication doses.
- It improves medication management and reduces handling errors.
This efficiency is crucial for getting products to market faster and managing complex trial logistics.
Achieve Corporate Sustainability Goals
Modern companies must demonstrate a commitment to sustainability. Automation helps pharmaceutical manufacturers achieve their environmental, social, and governance (ESG) goals. For example, some companies now use automation and analytics to transform their ESG reporting from dozens of spreadsheets into a single, consolidated report. This technology enables monthly updates on sustainability metrics, a major improvement over annual manual reporting. By integrating automation, companies can better track resource use and minimize waste, strengthening their corporate responsibility profile.
Adapt to Evolving Packaging Formats
Pharmaceutical packaging is rapidly changing. New trends include sustainable materials, smart features like QR codes, and enhanced security seals. A modern Deblister Machine is designed for this future.
These machines offer the flexibility to handle diverse packaging. They use adjustable settings and interchangeable parts to process various blister pack sizes, shapes, and materials, including both plastic and ALU-ALU formats.
This adaptability ensures that a company’s investment remains valuable as packaging technology continues to evolve, preventing the need for new equipment with every market shift.
Selecting the Right Deblister Machine for Your Needs
Choosing the right automation partner is a strategic decision. Companies must evaluate several factors to ensure their investment aligns with production needs, safety standards, and operational goals. A thorough assessment guarantees the selected equipment will deliver long-term value.
Assess Material and Pack Type Compatibility
A machine's primary function is to handle a company's specific product packaging. Compatibility is therefore the first point of evaluation. The equipment must process the full range of blister packs used in production. Decision-makers should create a checklist to confirm the machine’s capabilities.
- Blister Type: Can it handle push-thru, child-resistant, peelable, and multi-product packs?
- Blister Layout: Is it compatible with all existing and planned layouts?
- Maximum Pack Size: Does it accommodate packs up to 105mm wide and 140mm long?
Answering these questions ensures the Deblister Machine will not limit future packaging choices.
Prioritize Operator Safety and Ergonomics
Protecting employees is a top priority in any manufacturing environment. The best machines include built-in safety features to prevent accidents. Companies should inspect equipment for essential protective components. Safety guards must be securely attached and in proper working order. Advanced models also equip features like an electrical brake switch and a lock switch to enhance operator protection during use and maintenance. These features are not optional; they are fundamental to creating a safe and responsible workplace.
Evaluate Ease of Cleaning and Changeover Speed
Efficiency depends on more than just processing speed. Quick changeovers and simple cleaning procedures are critical for maintaining high throughput and compliance. Equipment should follow GMP sterile manufacturing design principles.
Surfaces made of stainless steel are smooth and non-reactive, which simplifies cleaning and prevents contamination.
Validated and repeatable cleaning processes are essential for passing FDA audits. Automated systems that document every batch help maintain certifications and ensure consistent quality. A machine that is easy to clean and quick to reconfigure minimizes downtime and maximizes productivity.
Delaying automation beyond 2026 creates a strategic liability. Some manufacturers already face significant bottlenecks from manual work, resulting in reduced productivity and higher operational costs. This inaction guarantees greater compliance risks and a loss of competitive standing.
Investing in an automatic deblister machine is not just an equipment upgrade. It is a fundamental business decision for future profitability and sustainability.
Companies must evaluate how this technology fits into their 2026 strategic plan. This proactive step will secure their operational future in an increasingly automated industry.
FAQ
What is the primary benefit of an automatic deblister machine?
An automatic deblister machine's primary benefit is cost reduction. It minimizes labor expenses and recovers nearly 100% of medication from blister packs. This process directly protects a company's bottom line and maximizes return on investment.
How does this machine improve regulatory compliance?
The machine improves compliance by creating secure, unalterable audit trails. It automatically logs batch data and operator actions. This meets FDA 21 CFR Part 11 requirements for data integrity, simplifying regulatory inspections and ensuring verifiable records.
Can the machine handle different types of blister packs?
Yes, modern deblister machines offer high flexibility. They use interchangeable parts and adjustable settings. This allows them to process various pack materials, sizes, and formats, including push-thru, child-resistant, and ALU-ALU packs, ensuring long-term operational adaptability.
What is the typical ROI for this equipment?
Companies often see a rapid return on investment (ROI), typically within 12 to 24 months. The ROI comes from significant labor cost reductions, near-total product recovery, and the elimination of expensive manual errors. This makes it a financially sound investment.
See Also
Exploring Cutting-Edge Folder Gluer Innovations for 2025 Carton Production Lines
Automatic UV Coaters: Indispensable for Achieving High-Speed, Flawless Print Finishes
Modern Capsule Decapsulators: Boosting Pharmaceutical Efficiency Through Advanced Technology
Comparing Leading Ampoule Filling Production Line Manufacturers: A Comprehensive Analysis
Essential Industrial Checkweighers: Optimizing Food Plant Operations and Quality Control