Your 2026 List of Top 5 Rotary Tablet Press Machine Makers
An excellent rotary tablet compression machine is crucial for pharmaceutical production. The pharmaceutical sector heavily depends on this technology, representing about 70% of the market. Selecting the right manufacturer is a key strategic decision. This guide offers a definitive 2026 ranking of the top five industry leaders. It details their core technologies and unique strengths. These insights will empower you to make an informed purchasing decision for your specific production needs.
#1. Fette Compacting: The Benchmark for High Performance
Company Overview
Fette Compacting has set industry standards for over 75 years. The company, founded around 1950, quickly became a pioneer in tablet production. It is now the world's leading provider of integrated solutions for industrial tablet manufacturing. A recent strategic partnership with CMIC in May 2025 reinforces its commitment to guiding clients from the laboratory to full-scale production. This long history of innovation establishes Fette Compacting as a benchmark for quality and performance in the pharmaceutical industry.
Core Technological Advantages
Innovation is central to Fette Compacting's market leadership. The company's engineering prowess is protected by a significant intellectual property portfolio.
Fette Compacting holds 140 patents. These patents cover core technologies for tablet production and continuous manufacturing.
Key innovations include a patented airflow system designed to prevent cross-contamination, a critical feature for maintaining product purity. Furthermore, its machines incorporate a patent-pending conical filling unit, which improves powder flow and tablet consistency. These proprietary technologies ensure superior operational efficiency and product quality.
Flagship Product Lines
The FE Series represents the pinnacle of Fette Compacting's engineering. This line includes the FE35 for rapid changeovers, the versatile FE55 for flexible production, and the high-output FE75 for large-scale manufacturing. The FE75 is a powerful double-sided Rotary Tablet Compression Machine capable of producing over 1.6 million tablets per hour. Its design incorporates the TRI.EASY concept for simple operation and servicing.
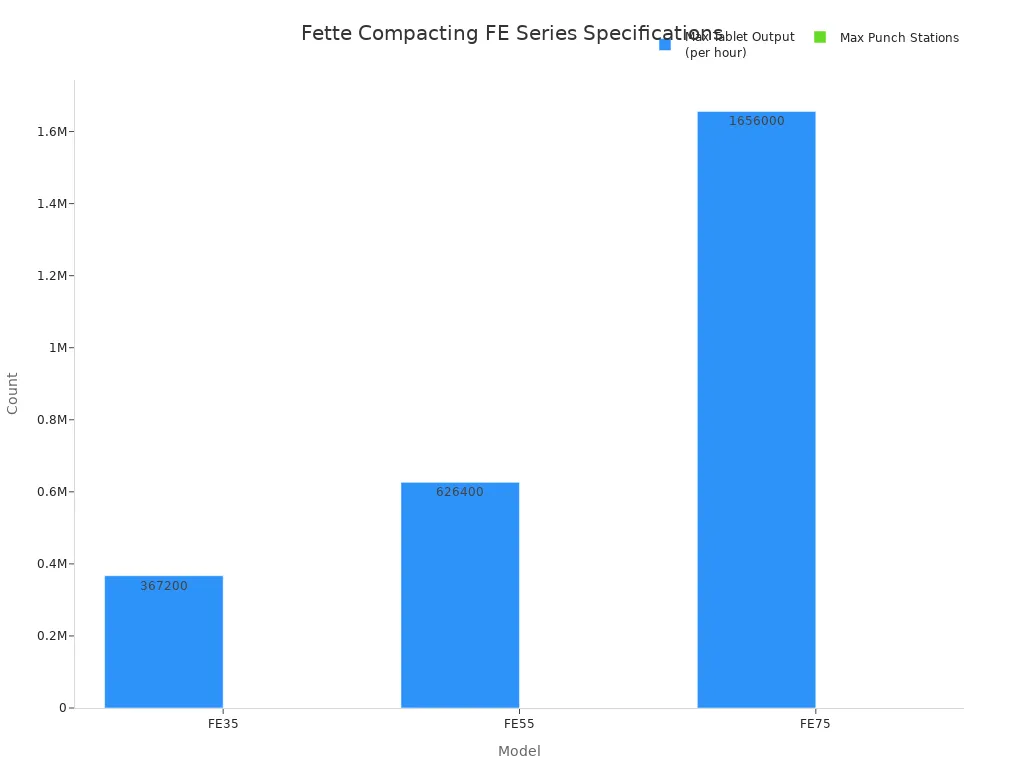
The table below details the specifications for each model in the FE series.
| Model | Max Tablet Output (per hour) | Max Punch Stations |
|---|---|---|
| FE35 | 367,200 | 51 |
| FE55 | 626,400 | 87 |
| FE75 | 1,656,000 | 115 |
These machines offer exceptional performance, reliability, and adaptability for modern pharmaceutical production.
Unique Selling Points
Fette Compacting distinguishes itself through a legacy of pioneering innovation and a commitment to providing complete, integrated production ecosystems. The company does not just sell machines; it delivers comprehensive solutions that encompass tablet presses, specialized tooling, and process equipment. This holistic approach ensures seamless integration and optimized performance from development to full-scale manufacturing. A long history of industry firsts, including the world's first computer-controlled press (PT2080) and the first press with an exchangeable turret (PT3090), solidifies its reputation as a technological leader.
Key differentiators that define Fette Compacting's market position include:
- Integrated Production Systems: The company offers a full suite of equipment, creating a unified and efficient manufacturing environment.
- Continuous Manufacturing (CM) Expertise: Its FE CPS system enables lean plant design, reduces process times, and facilitates rapid product changeovers for enhanced cost-efficiency.
- Next-Generation 'i Series': The F10i and F30i models deliver flexible, dust-tight, and digitally-ready production, setting a new standard for modern pharmaceutical operations.
- Unmatched Efficiency: The FE Series, particularly the FE35, is renowned for offering the industry's fastest product changeovers, minimizing downtime significantly.
For manufacturers focused on high-volume output, the FE75 model stands as a testament to Fette Compacting's engineering prowess. This machine is a production powerhouse.
The FE75 can produce over 1.6 million tablets per hour and press complex single-layer or double-layer tablets within a compact two-square-meter footprint.
Its design incorporates four compression rollers and up to 115 punch stations, allowing for exceptional control and throughput. Advanced features like pneumatically controlled tablet scrapers and a tubeless extraction unit further enhance its reliability and operational excellence, making it a premier choice for large-scale pharmaceutical production.
#2. KORSCH AG: Precision Engineering and Flexibility
Company Overview
KORSCH AG is a global leader celebrated for its precision engineering and cutting-edge tablet press technology. The company provides a comprehensive product line that includes granulation equipment, coating systems, and specialized tooling, focusing on tailored client solutions. A key strategic move was the acquisition of Medel Pharm in February 2023. This merger enhanced KORSCH's compaction simulator technology and R&D capabilities, combining over 135 years of experience in small-scale, fully instrumented tablet compression. This positions KORSCH as a versatile partner for projects from early development to full-scale production.
Core Technological Advantages
KORSCH excels at integrating smart technology into its machines, aligning with Industry 4.0 digitalization. Its presses utilize open-architecture control platforms, enabling seamless communication for predictive analytics and cloud computing. This advanced connectivity is a core strength.
KORSCH machines feature IO-Link smart sensors that transmit critical data for AI interpretation and predictive maintenance, optimizing overall equipment effectiveness (OEE).
Another key innovation is the PharmaView system. This system uses Microsoft HoloLens to provide augmented reality (AR) support for operators during setup, operation, and maintenance. This technology improves safety and reduces training time. The integrated PharmaControl system offers closed-loop feedback for precise tablet weight control, ensuring exceptional product consistency.
Flagship Product Lines
KORSCH offers a diverse portfolio designed for flexibility and performance. The X 3 and X 5 models are prime examples of smart tablet presses, featuring intuitive Smart-Touch HMIs for real-time data access and remote diagnostics. The X 5 MFP version is particularly notable for its ability to produce single-layer, bi-layer, and tri-layer tablets. The expanded KORSCH-MEDELPHARM R&D lineup serves every stage of development:
- XP 1: A robust single-punch press for small-batch production.
- STYL'One Evo: An advanced compaction simulator for multi-layer formulation development at production speeds.
- XL 100: A small-scale Rotary Tablet Compression Machine ideal for clinical batch production.
This range demonstrates KORSCH's commitment to providing solutions that scale with a client's needs, from initial research to commercial manufacturing.
Unique Selling Points
KORSCH AG establishes its market leadership through unparalleled flexibility, serving clients from early-stage research to high-volume commercial manufacturing. The company's primary strength lies in its ability to provide tailored solutions that grow with a project's needs. Its simple design philosophy and ease of operation make its machines a top choice for companies requiring responsiveness and versatility.
Key differentiators for KORSCH include:
-
Full-Spectrum Scalability: KORSCH offers a seamless path from development to production. It provides precise single-punch and efficient rotary presses for laboratory work. Mid-range tablet presses with 16-30 stations allow for effective product scale-up. For large-scale manufacturing, KORSCH delivers high-speed machines engineered for maximum precision and efficiency.
-
Advanced Process Optimization: The company equips its instrumented presses with sophisticated controls. These features allow operators to optimize precompression, main compression, and ejection forces during long production runs. Advanced systems offer electronic audit trails, press force control, and single-tablet rejection, which characterize production parameters and streamline technology transfer.
-
Material-Sparing R&D: KORSCH's compaction simulators are a game-changer for formulation development. They empower researchers to predict tablet performance at high speeds while using minimal amounts of valuable active pharmaceutical ingredients (APIs).
This capability is ideal for companies in the research and development phase or those focusing on innovative and high-potency drugs. It significantly reduces costs and accelerates the development timeline.
- Modular and Versatile Design: KORSCH machines are not limited to the pharmaceutical sector; their versatility extends to the food and chemical industries. The equipment features a modular design that simplifies maintenance and allows for future upgrades. This forward-thinking approach ensures a company's investment can adapt to changing production requirements, safeguarding its long-term value.
#3. GEA (Courtoy): Leaders in Continuous Manufacturing
Company Overview
GEA solidified its position in the pharmaceutical sector through the strategic acquisition of Courtoy, a historic leader in tablet compression. This move combined GEA's process engineering expertise with Courtoy's specialized equipment legacy. Today, Courtoy operates as a key brand within the GEA group, driving innovation for both established and emerging markets. The company stands as a major competitor in the high-speed tablet press market, leveraging decades of experience to deliver robust and reliable manufacturing solutions.
Core Technological Advantages
GEA's primary advantage is its pioneering role in continuous manufacturing (CM). The company's ConsiGma® platform is a fully integrated system that transforms powder into coated tablets within a single, contained production line. This technology offers significant benefits for modern pharmaceutical production.
- It directly links granulation and drying with tablet compression and coating.
- The system excels at processing challenging formulations, including high-drug-load tablets with "sticky" APIs.
- GEA's Conductor software provides seamless control over the entire process, ensuring complete data integrity and process transparency.
This integrated approach minimizes manual handling, reduces facility footprint, and accelerates production time from development to market.
Flagship Product Lines
The MODUL series represents the core of GEA's tablet press offerings. The line's central innovation is the Exchangeable Compression Module (ECM), a self-contained unit that includes all product-contact parts. This design isolates the compression zone from the mechanical parts of the press, enhancing containment and simplifying cleaning.
The ECM enables an exceptionally fast product changeover, often completed in just 30 minutes. This feature dramatically reduces downtime and boosts overall equipment effectiveness (OEE).
The series offers a machine for every production scale. The MODUL P is designed for R&D and clinical batch production, ensuring seamless scale-up. For full-scale manufacturing, the MODUL Q provides high efficiency in a compact footprint. The high-output MODUL D and MODUL S models cater to super-high volume applications and the production of complex bilayer tablets, making the MODUL line a versatile solution for any Rotary Tablet Compression Machine requirement.
Unique Selling Points
GEA distinguishes itself through its unmatched expertise in fully integrated continuous manufacturing (CM). The company provides end-to-end solutions that transform raw materials into finished tablets within a single, contained production line. This holistic approach, backed by deep process knowledge, builds significant customer confidence and sets GEA apart from competitors who focus solely on individual machines.
GEA has been a recognized leader in continuous processing since introducing its ConsiGma™ system in 2008. Its global competence center and extensive experience in batch granulation, drying, and compression technologies provide a strong foundation for its market leadership.
The company's solid dosage production platform is a testament to this integrated philosophy. It combines multiple advanced technologies into one seamless system. Key components include:
- Continuous blending, wet or dry granulation, and drying processes.
- The high-speed Courtoy™ MODUL™ tablet press with an integrated tablet relaxation module.
- The first commercially available continuous GEA coater for final product finishing.
- Advanced Process Analytical Technology (PAT) instruments, such as the patented Lighthouse Probe™, for real-time quality monitoring.
A cornerstone of this system is the Exchangeable Compression Module (ECM). The ECM contains all product contact parts and is completely isolated from the rest of the press. Operators can easily remove the module for offline washing, which keeps the main press body powder-free and eliminates cleaning downtime. This design provides excellent containment for high-potency APIs and allows for rapid product changeovers. This integrated control system enables the 'real-time-release' of finished tablets, accelerating production and ensuring consistent quality.
#4. IMA Group (Kilian): Robust Design and Versatility
Company Overview
IMA Group's solid dose division includes the esteemed Kilian brand, a name synonymous with industrial strength. Kilian has a rich history in designing and manufacturing high-performance equipment. The brand is globally recognized for its robust, durable, and long-lasting tablet presses. Kilian's engineering team successfully merges traditional craftsmanship with modern technology. This combination results in machinery built for high-production environments that demand optimal efficiency and precision.
Core Technological Advantages
Kilian's design philosophy prioritizes robustness and high yield. Its presses often feature a 'punches up' configuration, a design that minimizes sampling delays and improves overall output. This focus on mechanical excellence ensures reliability during long production runs.
Many Kilian presses, such as the Romaco Kilian RX series, also integrate sophisticated force monitoring systems. This technology provides real-time quality control, ensuring tablet consistency and adherence to specifications.
These core principles make Kilian machines a trusted choice for manufacturers who require dependable performance and minimal downtime.
Flagship Product Lines
The KILIAN KTP series is the cornerstone of the company's offerings, engineered for versatility and power. These machines excel at handling difficult formulations, including sticky or temperature-sensitive products, and are designed to prevent common issues like capping. The KTP 420X, for example, can produce mono-layer, bilayer, or core tablets, demonstrating its adaptability. This powerful Rotary Tablet Compression Machine is a testament to Kilian's engineering.
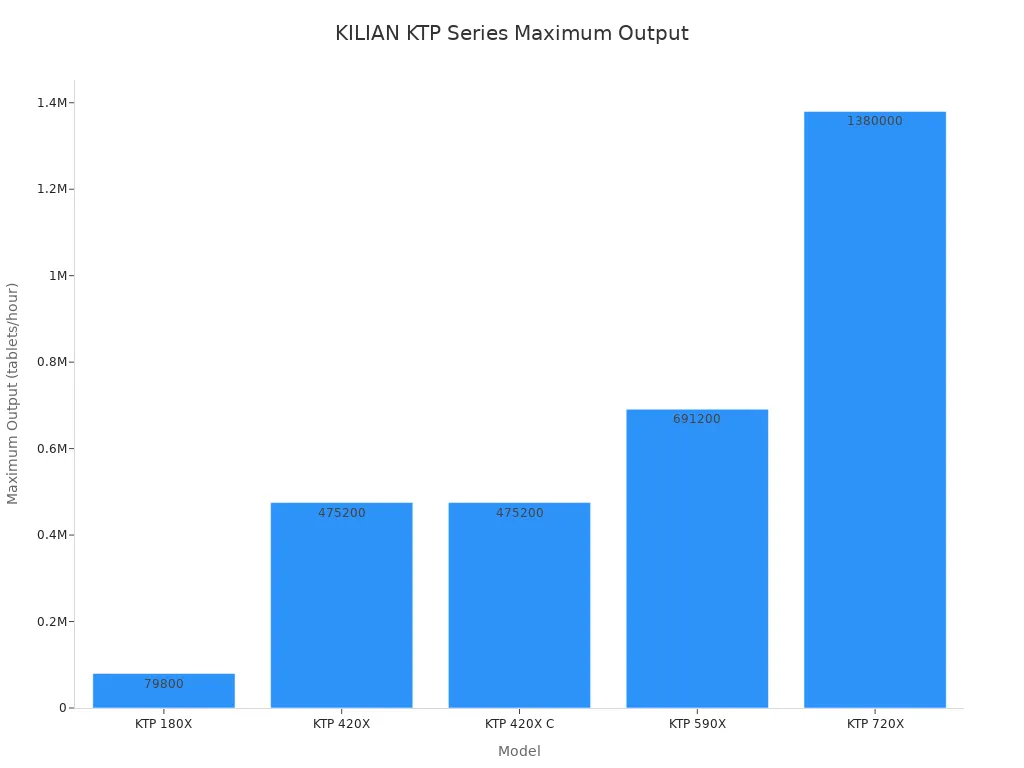
The series offers a range of outputs to meet diverse production scales.
| Model | Maximum Output (tablets/hour) |
|---|---|
| KTP 420X | 475,200 |
| KTP 590X | 691,200 |
| KTP 720X | 1,380,000 |
This lineup ensures that whether for mid-size or massive-scale production, there is a Kilian press built for the task.
Unique Selling Points
IMA Kilian builds its market reputation on a foundation of German engineering, focusing on robustness, durability, and a low total cost of ownership. The brand delivers machines that are not just powerful but also exceptionally reliable, making them a long-term asset for any high-volume production facility. Kilian's equipment is recognized for its precision, efficiency, and straightforward operation.
The company's design philosophy emphasizes both performance and practicality. This focus on user-centric engineering simplifies maintenance and maximizes uptime. Key design features include:
- Effortless Cleaning: All components have smooth surfaces. Operators can remove the die table without detaching the punches.
- Easy Access: The force feeder and tablet discharge are hinged. They swing out to provide clear access for cleaning and inspection.
- Superior Contamination Control: Kilian completely separates and seals the mechanical and compression areas. This design prevents product from entering the lower machine compartment, reducing cross-contamination risks and maintenance needs.
This meticulous attention to detail ensures that Kilian presses are not only powerful but also remarkably easy to manage.
Kilian machines are an investment in operational continuity. Their robust construction minimizes wear and tear, while the smart design reduces cleaning time. This combination results in less downtime and a lower total cost of ownership over the machine's long service life.
For manufacturers prioritizing industrial strength, high reliability, and operational efficiency, Kilian offers a compelling value proposition. The brand's commitment to building durable, user-friendly machinery ensures consistent performance in the most demanding production environments.
#5. Syntegon: Integrated Solutions and Reliability
Company Overview
Syntegon, formerly Bosch Packaging Technology, rebranded in 2020 and has since solidified its role as a strategic lifecycle partner for the pharmaceutical and food industries. With over 160 years of experience, the company leverages a rich history of innovation. Key milestones include the 2023 joint venture with Klenzaids and the 2024 acquisition of Telstar, which expanded its process technology portfolio. Syntegon supports approximately 75,000 installed systems globally. It generated 1.6 billion euros in sales in fiscal year 2024, underscoring its position as a major force in providing technologies for safe and efficient medicine production.
Core Technological Advantages
Syntegon's primary strength lies in its ability to deliver fully integrated, end-to-end solutions. The company acts as a full-service provider, aiming for fewer interfaces and seamless integration of mission-critical processes. This holistic approach ensures reliability and enhances productivity for its customers.
Syntegon leverages deep engineering expertise to provide innovative and high-performance technology. This strategy helps customers unlock their full potential by creating a unified and efficient production environment from a single source.
Their comprehensive service portfolio covers the entire machine lifecycle. Services range from spare parts management to digital line optimization, ensuring smooth production and high uptime.
Flagship Product Lines
Syntegon offers a robust portfolio of processing and packaging technology. The TPR series of tablet presses is known for its reliability and efficiency in demanding production environments. These machines are designed for high performance and easy integration into larger production lines. The TPR 200, for example, is a versatile Rotary Tablet Compression Machine suitable for small to medium batch sizes.
Beyond tablet presses, Syntegon's expertise in integrated systems is evident in products like the Versynta Flexible Filling Platform (FFP). This system uses pre-developed and tested modules that can be flexibly combined, including integrated isolator technology. Furthermore, its subsidiary Pharmatec supplies pure media solutions, providing all necessary components for a complete system infrastructure. This single-source approach simplifies project management and ensures all parts work together seamlessly.
Unique Selling Points
Syntegon differentiates itself by acting as a comprehensive lifecycle partner, offering end-to-end solutions that extend far beyond the initial machine sale. The company provides a single source for integrated systems, ensuring seamless project management and operational reliability. This holistic approach builds customer confidence and simplifies the entire production process. Syntegon’s commitment to long-term partnership is evident in its extensive service portfolio.
This support structure is designed to maximize uptime and future-proof a manufacturer's investment. Key service offerings include:
- Expert engineering and qualification for new lines and projects.
- Flexible service agreements covering maintenance, parts, and technical support.
- Scalable modernizations and upgrades to meet new performance requirements.
- Digital solutions for real-time data monitoring and predictive maintenance.
- Tailored training programs for operators and maintenance staff.
A core component of this digital strategy is Synexio. This cloud-based software solution collects and visualizes critical equipment data. It provides data-driven insights that enhance transparency, optimize equipment performance, and reduce unplanned downtime.
This digital integration allows Syntegon to offer powerful remote services. Experts can provide immediate assistance through a secure connection for troubleshooting, software backups, and parameter reviews. This capability significantly reduces travel costs and shortens reaction times, minimizing production interruptions. For manufacturers seeking a reliable partner who provides integrated systems and robust, lifelong support, Syntegon presents a compelling and secure investment.
How to Choose the Right Rotary Tablet Compression Machine
Selecting the ideal machine requires a careful evaluation of production needs, formulation characteristics, and regulatory obligations. A strategic choice aligns the equipment's capabilities with your operational goals.
Match Machine to Production Scale
Manufacturers must first align the machine's output capacity with their production targets. A mismatch can lead to either costly underutilization or an inability to meet market demand. Production scale directly influences the required machine type.
| Production Scale | Machine Type | Output Range (tablets/hour) |
|---|---|---|
| Small-scale | Low-speed small-scale rotary press | 13,200 to 37,800 |
| Mid-scale | 16-station machines | 100,000 to 400,000 |
| Large-scale | High-speed rotary press | Up to 960,000+ |
The number of punch stations on a turret significantly impacts production volume. For example, a 55-station press can more than triple the output of a 16-station press running at the same speed. While larger turrets boost capacity, they also require more sophisticated controls and increase maintenance demands.
Consider Formulation Complexity
The physical properties of the powder formulation are critical. Variables like particle size, shape, and flowability directly affect manufacturing success. Most powders need granulation to achieve the flow rates necessary for high-speed tableting. A machine must be able to handle the specific characteristics of your product.
Complex products, such as multi-layer or core-coated tablets, demand specialized features. Key capabilities include:
- High-precision spray control for accurate layering.
- Advanced layering systems to prevent cross-contamination.
- Integrated drying systems to ensure smooth layer adhesion.
- Intelligent controls for recipe storage and real-time monitoring.
Ensure Regulatory Compliance
Adherence to industry regulations is non-negotiable. Equipment used in drug manufacturing must comply with Good Manufacturing Practices (GMP) and other standards. Modern tablet presses are engineered to meet these requirements through extensive data logging and validated cleaning procedures.
A crucial standard is FDA 21 CFR Part 11, which governs electronic records and signatures. This regulation mandates that a machine's control system provides secure, time-stamped audit trails for all actions. Essential features include real-time logging of process parameters, electronic signatures for batch records, and detailed logs of data access. These functionalities ensure data integrity and traceability for every batch.
Analyze Total Cost of Ownership
A comprehensive financial evaluation extends beyond the initial purchase price. Manufacturers must analyze the total cost of ownership (TCO) to understand the long-term financial impact of the equipment. This analysis provides a realistic budget for the machine's entire service life. The initial investment is only one part of a larger financial picture. A complete TCO calculation includes multiple direct and indirect expenses.
Key factors contributing to the total cost of ownership include:
- Initial equipment cost
- Installation, qualification, and validation expenses
- Cost of spare parts and tooling, such as punches and dies
- Preventive maintenance and potential repair costs
- Labor costs for operators and mechanics
- Energy consumption, including compressed air or vacuum
- Costs incurred due to downtime when the press is not operational
Running costs, like electricity consumption and wear on parts, are crucial long-term budget considerations. Energy-efficient models can significantly lower operational expenses over time. Tooling also represents a major investment, as manufacturers often purchase multiple sets during a product's lifecycle. A consistent maintenance schedule is essential for optimal performance and machine longevity.
Proactive maintenance is critical for tooling. Proper lubrication, cleaning, polishing, and storage extend tool life significantly. This practice reduces variable costs and helps maintain high production output. It ultimately maximizes the return on investment by lowering long-term expenses.
Poorly maintained tooling leads to frequent replacements and can compromise tablet quality. By contrast, a well-managed maintenance program protects the investment, minimizes downtime, and ensures consistent, high-quality production for years to come.
Selecting a manufacturer—Fette, KORSCH, GEA, IMA, or Syntegon—is a critical strategic decision. The ideal partner aligns with a company's unique production requirements. A successful choice depends on a careful evaluation of several key factors:
- Production capacity and scale
- Formulation complexity and challenges
- Long-term budget and total cost of ownership
This guide provides a checklist to help manufacturers evaluate vendors. It empowers them to secure the best Rotary Tablet Compression Machine for their facility.
FAQ
What is the main difference between a single-punch and a rotary press?
A single-punch press uses one set of tooling to produce tablets one at a time, ideal for R&D. A rotary press uses multiple tooling stations on a rotating turret. This design allows for continuous, high-speed production suitable for large-scale manufacturing.
Why is tooling so important for a tablet press?
Tooling (punches and dies) directly shapes the final tablet. Its quality affects tablet weight, hardness, and appearance. Proper tooling maintenance is critical. It prevents product defects, reduces machine wear, and ensures consistent, high-quality output, maximizing the return on investment.
What does 'continuous manufacturing' mean for tablet production?
Continuous manufacturing (CM) integrates multiple production steps into one seamless process.
For example, a CM line can combine powder blending, granulation, compression, and coating without interruption. This approach reduces production time, minimizes facility footprint, and enhances overall process control and efficiency.
How does a company choose between these top manufacturers?
A company selects a manufacturer based on specific needs. Key factors include:
- Required production volume (R&D vs. large-scale)
- Complexity of the tablet formulation
- Budget and total cost of ownership
- Need for integrated systems or standalone machines
Each brand offers unique strengths for different operational priorities.
See Also
Essential Folder Gluer Innovations for 2025 Carton Packaging Production
Top 5 Applications for Single-Stage Rotary Vane Vacuum Pump Sets
Six Must-Have Industrial Vacuum Cleaners for Every Modern Business
Three Essential Industrial Checkweighers for Optimal Food Plant Operations