Syringe inspection x-ray: can it really catch hidden dangers?
Syringe inspection x-ray offers manufacturers a reliable method to detect hidden dangers in syringes. This advanced technology identifies defects that escape visual checks.
Patients rely on safe medical devices. X-ray inspection reveals issues invisible to the naked eye, helping ensure syringe safety and quality control.
How Syringe Inspection X-ray Works and What It Detects
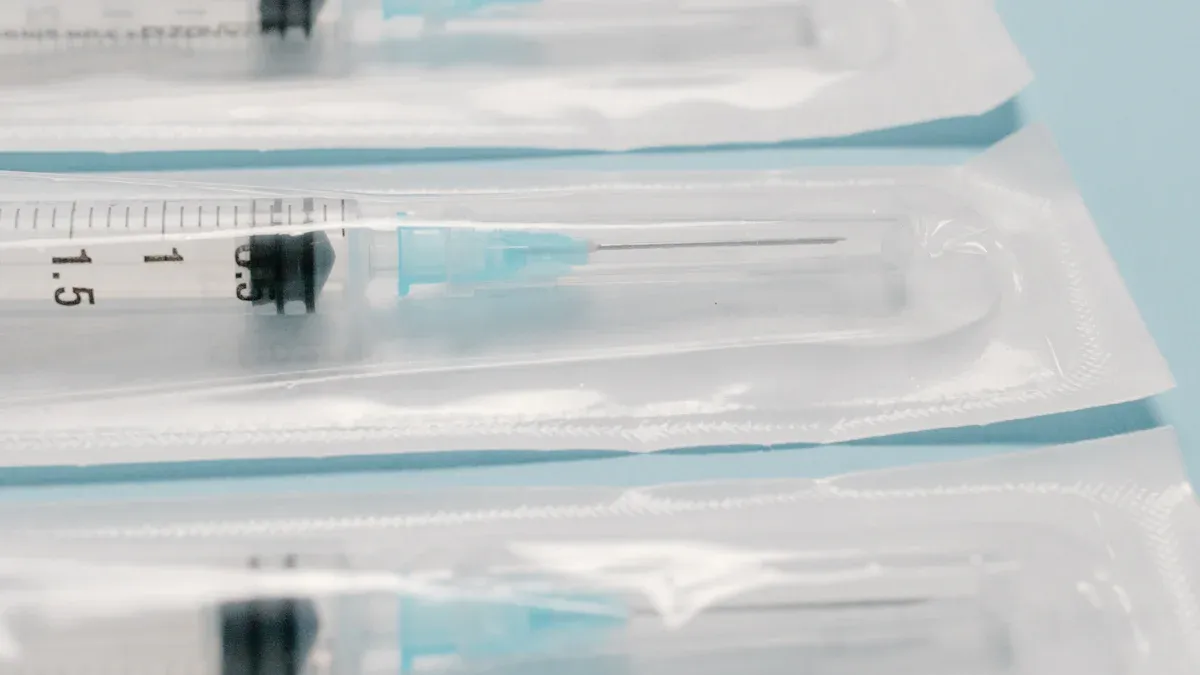
How X-ray Technology Inspects Syringes
Syringe inspection x-ray relies on advanced imaging to visualize the internal structure of syringes. The technology uses greyscale imaging, where darker areas on the x-ray image indicate more material in the beam path. This method allows for detailed analysis of each syringe, revealing dimensional discrepancies such as syringes that are too short or too long. The system can inspect sealed packaging and verify the presence of all components, even those hidden from view.
X-ray inspection systems count syringes and needles inside packaging, ensuring that every kit contains the correct number of items. This process supports manufacturers in meeting strict quality control standards.
The most common x-ray systems used in the medical device industry include:
| X-Ray System Type | Common Applications |
|---|---|
| Digital Realtime X-ray (DR) | Failure analysis, product development, metrology, first-article inspection |
| Computed Tomography (CT) | Quality assurance for syringes and other medical devices |
These systems integrate with automated manufacturing lines, providing non-destructive testing that detects contaminants and verifies fill levels. Although integration requires customization and may increase costs, the benefits for product integrity are substantial.
Detecting Hidden Defects: Cracks, Air Bubbles, and Foreign Particles
Syringe inspection x-ray excels at identifying defects invisible to the naked eye. The technology detects cracks in syringe barrels, air bubbles trapped within the liquid, and foreign particles that could compromise sterility. Manufacturers use advanced algorithms to improve detection accuracy, generating reports that estimate the percentage of defective syringes and the likelihood of false negatives.
- Inspection machines produce detailed production reports.
- Experts manually verify samples to validate machine data.
- High precision and recall values help balance defect detection, minimizing false negatives and avoiding excessive false positives.
Industry standards require x-ray inspection to ensure package integrity and count components in disposable syringe kits. The process detects damaged parts, reducing the risk of product recalls and enhancing patient safety.
X-ray inspection technology is vital in the pharmaceutical industry. It ensures products are free from contaminants and that all necessary components are present, aligning with industry standards for quality control.
Identifying Assembly Errors and Needle Misalignment
Assembly errors and needle misalignment pose significant risks in syringe manufacturing. Syringe inspection x-ray identifies these issues by analyzing the internal structure and alignment of components. The technology can spot needles that are not properly seated or are bent, which could lead to malfunction or injury.
- X-ray systems verify that critical components are present and correctly assembled.
- The process prevents potential patient injuries by ensuring proper alignment.
- Manufacturers adhere to stringent regulations, using x-ray inspection to enhance product safety and compliance.
Safety considerations remain a priority. Pulsed x-ray technology reduces radiation exposure to levels far below regulatory thresholds. Simple shielding methods suffice, making integration into production lines straightforward and safe for workers.
Future advancements in syringe inspection x-ray focus on improved detection of internal flaws, foreign particles, and fill inconsistencies. AI-powered imaging and machine learning algorithms promise to revolutionize defect detection, while 3D tomography emerges as a significant technology in this field.
Syringe Inspection X-ray vs. Other Quality Control Methods
Visual and Manual Inspection: What Gets Missed
Visual and manual inspection remain common in syringe manufacturing, but these methods have significant limitations. Human inspectors often miss subtle defects due to performance variability and fatigue. The probability of detecting a 150-micron particle hovers around 70%, and smaller particles escape notice even more frequently. Inspectors show variability in their detection rates, both between individuals and over time. Bias in defect classification can also affect reliability. Expecting 100% detection of critical defects is unrealistic and introduces systemic risks.
- Human performance varies, leading to inconsistent results.
- Not all defects are found due to the probabilistic nature of detection.
- Bias and fatigue reduce reliability.
Unique Advantages of X-ray Inspection
Syringe inspection x-ray offers several advantages over traditional methods. The technology processes hundreds of syringes per hour, providing high accuracy and nondestructive testing. It detects functional defects, such as needle misalignment, that manual inspection may overlook. The following table highlights key differences:
| Advantage | Syringe Inspection X-Ray | Traditional Methods |
|---|---|---|
| Speed | Processes hundreds/hour | Slower processing rates |
| Accuracy | High accuracy | Variable accuracy |
| Nondestructive Testing | Yes | Often destructive |
| Functional Defect Detection | Yes | Limited capabilities |
X-ray inspection also surpasses other technologies in speed and effectiveness, identifying a wider range of contaminants and defects than methods focused solely on metal detection.
Real-World Effectiveness and Limitations
Syringe inspection x-ray has transformed quality control in the medical device industry. Automated inspection machines reduce human error and improve operational efficiency, lowering recall risks and supporting regulatory compliance. Regulatory mandates and FDA standards drive adoption, while increased consumer awareness fuels demand for product safety. Manufacturers benefit from cost-effective third-party x-ray inspection, which is far less expensive than a full product recall.
Currently, the one major limitation of X-ray imaging is that it is not ideally suited yet for in-line analysis, particularly for certain features of interest. The technology is constantly evolving, though, with higher-resolution and higher-speed detectors, in combination with advancements in software analysis, which will help overcome the issues that are limiting the use of X-ray analysis in production environments.
- Syringe inspection x-ray provides manufacturers with a reliable method to detect hidden dangers in syringes.
- This technology reduces the risk of defective syringes reaching patients.
- Manufacturers rely on it as a critical part of modern syringe quality assurance.
FAQ
What types of defects can syringe inspection x-ray detect?
Syringe inspection x-ray identifies cracks, air bubbles, foreign particles, and needle misalignment. The system also verifies assembly errors and missing components.
Is syringe inspection x-ray safe for workers?
Manufacturers use pulsed x-ray technology. Shielding methods keep radiation exposure well below regulatory limits. Workers remain safe during routine operation.
How does syringe inspection x-ray compare to manual inspection?
- X-ray inspection provides higher accuracy and speed.
- Manual inspection often misses subtle defects due to human error and fatigue.
See Also
Top Locations for Purchasing X-Ray Inspection Equipment in China
Is It Worth Investing in Automated Virus Sampling Lines?
Understanding Pharmaceutical Checkweighers and Their Key Characteristics