Essential Steps to Achieve PED 2014/68/EU Certification
Manufacturers achieve PED 2014/68/EU certification by following strict procedures for their product. The pressure equipment directive 2014/68/eu applies to pressure equipment like plate heat exchangers from SHPHE. Compliance enables entry into the EU market, ensures safety, and fulfills legal requirements.
| Benefit/Requirement | Description |
|---|---|
| Access to EU Market | Compliance with PED is necessary to enter the EU pressure equipment market. |
| Revenue Potential | Achieving PED compliance can lead to increased revenue opportunities. |
| Legal Protection | Compliance helps avoid legal issues related to nonconformities. |
| Safety Assurance | Ensures that pressure equipment meets established safety standards. |
Identify If Your Product Is Subject to Pressure Equipment Directive 2014/68/EU
Types of Pressure Equipment Covered (Including SHPHE and Plate Heat Exchangers)
Manufacturers must first determine if their product falls under the scope of the pressure equipment directive 2014/68/eu. The directive applies to a wide range of pressure equipment, including plate heat exchangers produced by Shanghai Heat Transfer Equipment Co., Ltd. (SHPHE). The following table outlines the main criteria that decide if a product is subject to the directive:
| Criteria | Description |
|---|---|
| Maximum Allowable Pressure | Equipment with a maximum allowable pressure greater than 0.5 bar is covered. |
| Scope of Equipment | Includes vessels, piping, safety accessories, and other pressure accessories. |
| Compliance Requirements | Manufacturers must meet essential safety requirements for design and manufacturing. |
Products such as plate heat exchangers, pressure vessels, and safety accessories often require certification under the pressure equipment directive 2014/68/eu. These products must meet strict safety and design standards before entering the EU market.
Exemptions and Special Cases Under PED 2014/68/EU
Not all pressure equipment requires certification. The pressure equipment directive 2014/68/eu lists several exemptions and special cases. Products that fall into these categories do not need to follow the full certification process:
- Pipelines for fluid transport to or from installations
- Water supply and distribution networks
- Simple pressure vessels under Directive 2014/29/EU
- Aerosol dispensers under Council Directive 75/324/EEC
- Equipment for vehicles defined by specific legal acts
- Equipment classified as no higher than category I under Article 13
- Items for nuclear use
- Well-control equipment in petroleum and gas industries
- Equipment where pressure is not a significant design factor, such as engines and turbines
- Blast furnaces and related systems
- High-voltage electrical equipment enclosures
- Pressurized pipes for transmission systems
- Ships, rockets, and aircraft
- Flexible casing pressure equipment like tires and inflatable crafts
- Bottles for carbonated drinks
- Vessels for transporting drinks with specific pressure limits
- Equipment covered by directives for dangerous goods
- Radiators and pipes in warm water heating systems
- Vessels containing liquids with low gas pressure
Note: Manufacturers should always review the directive carefully to confirm if their product qualifies for an exemption before proceeding with certification.
Classify Your Product According to PED 2014/68/EU Categories
Product Categories and Groups Explained
Manufacturers must classify each product under the pressure equipment directive 2014/68/eu before starting the certification process. The directive divides pressure equipment into specific categories and groups based on design, intended use, and risk profile. Each pressure device or assembly falls into a category that determines the level of scrutiny required for certification. The following list outlines the main groups recognized by the directive:
- Equipment for nuclear use, where failure may cause radioactivity emission.
- Well-control equipment for petroleum, gas, or geothermal extraction.
- Pressure device or assembly designed with strength and stability as primary factors.
- Blast furnaces and related systems.
- Enclosures for high-voltage electrical equipment.
- Pressurized pipes for transmission systems.
- Ships, rockets, aircraft, and mobile offshore units.
- Pressure equipment with flexible casings.
- Exhaust and inlet silencers.
- Bottles or cans for carbonated drinks.
- Vessels for drink transport and distribution with specific pressure limits.
- Equipment covered by other directives or international codes.
- Radiators and pipes in warm water heating systems.
- Vessels for liquids with limited gas pressure.
Tip: Proper classification ensures that each product meets the correct requirements for pressure equipment directive 2014/68/eu certification.
Determining Hazard Levels for Certification
Manufacturers must assess the hazard level of each pressure device or assembly to comply with ped 2014/68/eu. The process involves several steps:
- Evaluate the fluid type and hazards. Group 1 includes hazardous fluids such as explosive, toxic, or highly flammable substances. Group 2 covers benign fluids.
- Assess stored energy and pressure. Review the stored energy in vessels or the pressure and diameter in pipes to determine risk.
- Determine the classification category. Assign the product to one of four PED categories (I-IV) based on complexity and risk.
- Select the appropriate conformity module. Choose the module that matches the product’s risk and category for pressure equipment directive 2014/68/eu certification.
Each step helps manufacturers identify the correct path for certification and ensures that every pressure device or assembly meets EU safety standards.
Determine the Applicable PED 2014/68/EU Conformity Assessment Procedure
Overview of Assessment Modules for Pressure Equipment
Manufacturers must select the correct conformity assessment procedures when seeking certification for a product under the pressure equipment directive 2014/68/eu. The procedure ensures that every product meets the requirements of ped 2014/68/eu and relevant harmonized standards applied. The assessment modules provide a structured approach for verifying compliance. Each module addresses different aspects of the product’s lifecycle, including design, manufacturing, testing, and final acceptance.
- Certified quality assurance systems oversee the design and production of pressure equipment.
- Notified bodies monitor compliance, review technical documentation, conduct audits, and confirm CE marking.
- Manufacturers attach the CE marking to each product, with the notified body responsible for oversight.
Harmonized standards play a critical role in these procedures. They guide manufacturers in meeting technical requirements and safety benchmarks for pressure equipment. Applying harmonized standards simplifies the certification process and demonstrates conformity with EU regulations.
Selecting the Right Procedure for Your Product
Choosing the appropriate conformity assessment procedure depends on the product’s classification and risk category. The following table outlines the recommended modules for each category of pressure equipment:
| Category of Equipment | Conformity Assessment Procedure |
|---|---|
| Category I | Module A |
| Category II | Module B |
| Category III | Module C |
| Category IV | Module D |
Manufacturers must evaluate the product’s hazard level and intended use before selecting a procedure. Harmonized standards ensure that the product meets all technical and safety requirements. The correct conformity assessment procedures streamline certification and support market access in the EU.
Tip: Always verify that harmonized standards align with the product’s design and application to ensure successful certification.
Prepare Technical Documentation for PED 2014/68/EU Certification
Required Documents Checklist for Pressure Equipment
Manufacturers must gather specific documents to meet the requirements of PED 2014/68/EU certification. Each product must have a complete set of records that demonstrate compliance with the pressure equipment directive 2014/68/eu. The checklist below outlines the essential requirements for technical documentation:
- Design drawings and specifications for the product
- Manufacturing process descriptions
- Material certificates and traceability records
- Test reports and inspection certificates
- Risk assessment documentation
- Declaration of conformity
- Operating instructions and maintenance guidelines
- Quality control and assurance records
These requirements help manufacturers prove that each product meets the safety and performance standards set by the EU. Proper documentation supports the certification process and ensures that pressure equipment can enter the market without delays.
Tip: Organize all documents in a clear and logical order to simplify review by notified bodies.
Key Elements to Include in Your Technical File
A technical file must contain several key elements to satisfy the requirements for PED 2014/68/EU certification. The table below summarizes the most important components:
| Key Element | Description |
|---|---|
| Declaration of Conformity | A document that states the equipment meets the necessary standards. |
| Technical Documentation | Detailed information about the design and manufacturing of the equipment. |
| Risk Assessment Records | Documentation that evaluates potential risks associated with the equipment. |
Manufacturers must ensure that each product’s technical file addresses all requirements. The technical documentation should describe the design, materials, and manufacturing processes. Risk assessment records must identify hazards and outline measures to control them. The declaration of conformity confirms that the product complies with the pressure equipment directive 2014/68/eu and meets EU standards.
Note: Regularly update the technical file to reflect changes in product design or manufacturing requirements.
Engage with a Notified Body for PED 2014/68/EU Certification
When a Notified Body Is Required for Your Product
Manufacturers must determine if their product requires assessment by a Notified Body to meet the requirements of PED 2014/68/EU. The pressure equipment directive 2014/68/eu outlines specific conditions where engagement with a Notified Body becomes mandatory. The following table summarizes these situations:
| Condition | Description |
|---|---|
| Directive or Regulation | The product falls under a directive or regulation requiring third-party assessment. |
| High-Risk Classification | The product is classified as high-risk and self-certification is not allowed. |
| Conformity Assessment | The applicable conformity assessment procedure requires a Notified Body review. |
Products such as plate heat exchangers often fall into categories where third-party review is essential. Manufacturers must review the requirements for each product to ensure compliance. Notified Bodies provide independent verification, which strengthens the certification process and supports market access in the EU.
Note: Failing to engage a Notified Body when required can result in non-compliance and prevent the product from entering the EU market.
How to Choose and Work with a Notified Body in the EU
Selecting the right Notified Body is a critical step in the certification journey. Manufacturers should evaluate several factors before making a decision:
- Review the Notified Body’s experience with pressure equipment and PED 2014/68/EU requirements.
- Confirm accreditation and recognition within the EU.
- Assess responsiveness and communication during the certification process.
- Request references for similar product types.
Once selected, manufacturers must provide complete technical documentation and respond promptly to requests for additional information. Clear communication helps ensure that the product meets all requirements and passes certification without delays.
Tip: Early engagement with a Notified Body can streamline the process and reduce the risk of non-compliance.
Complete Product Testing and Quality Assurance for PED 2014/68/EU
Testing Requirements for Pressure Equipment
Manufacturers must ensure that every product meets the essential safety requirements set by the pressure equipment directive 2014/68/eu. Testing plays a vital role in confirming that each product complies with safety standards before entering the EU market. The directive outlines specific requirements for different material types used in pressure equipment. The table below summarizes the main testing requirements:
| Material Type | Requirements |
|---|---|
| Carbon Steels | Must demonstrate a ductility level of 14% and impact resistance of 27J at 20°C or lowest operating temperature. |
| Ferritic Steels | Same requirements as carbon steels. |
| Nickel Alloy Steels | Must meet the same ductility and impact resistance criteria. |
| ASME Grades | Specific material appraisals required; must meet SA/SB requirements and impact properties. |
| PMA | One-time evaluation and approval process for materials, requiring submission to a notified body. |
Manufacturers must document all test results and keep records for certification. These steps help ensure that the product meets the strict requirements for pressure equipment and aligns with EU safety standards.
Quality Management Systems and Certification
A robust quality management system supports consistent compliance with PED 2014/68/EU. The most recognized system is ISO 9001, which many industries use to manage quality. Notified bodies may also issue PED-specific certificates to material suppliers, confirming that their products meet the requirements for certification. The table below highlights the main quality management systems:
| Quality Management System | Description |
|---|---|
| ISO 9001 | Commonly relied upon for quality management in various industries. |
| PED-specific certificates | Issued by notified bodies for material suppliers to ensure compliance with PED requirements. |
Manufacturers should note that not all ISO registrars can audit PED systems. Certification bodies such as TÜV Rheinland provide both product and system certification services according to PED 2014/68/EU. These systems help manufacturers maintain high standards and meet all essential safety requirements throughout the product lifecycle.
Draft and Maintain the EU Declaration of Conformity for PED 2014/68/EU
Content and Format Requirements for the Declaration
Manufacturers preparing the eu declaration of conformity for pressure equipment under PED 2014/68/EU must follow strict guidelines. The document serves as official proof that the product meets all requirements of the pressure equipment directive 2014/68/eu. Each ce declaration of conformity must include essential information to ensure traceability and compliance. The following table outlines the main content and format requirements:
| Requirement | Description |
|---|---|
| Manufacturer Name and Address | The name and address of the manufacturer must be included. |
| Product Identification | Product name, type, model, and serial or batch number must be specified. |
| CE Markings | List the CE markings claimed for the equipment. |
| Harmonized EN Standards | Indicate the harmonized EN standards used for design and evaluation. |
| Compliance Statement | Include a statement confirming compliance with the Directive(s). |
| Signature | A company officer must sign the document. |
| Additional Items | Optional: test report numbers, third-party lab details. |
A clear and complete eu declaration of conformity supports the certification process and demonstrates that the product meets all legal and safety standards in the EU.
Responsibilities for Updates and Recordkeeping
Manufacturers hold ongoing responsibilities for maintaining the eu declaration of conformity throughout the product lifecycle. They must update the ce declaration of conformity if the product design changes or if new standards apply. The table below summarizes these responsibilities:
| Responsibility | Details |
|---|---|
| Design and Manufacture | Ensure compliance with essential safety requirements in Annex I. |
| EU Declaration of Conformity | Draw up as per ANNEX IV, providing conformity information. |
| CE Marking | Affix the CE marking to the equipment or assembly. |
| Series Production Considerations | Account for design changes and updates to standards. |
| Implementation Actions | Conduct sample testing, maintain complaints register, inform distributors of corrective actions. |
Manufacturers must keep records of each eu declaration of conformity and related certification documents. Regular reviews help ensure that every product remains compliant with PED 2014/68/EU and supports safe use of pressure equipment in the EU market.
Apply CE Marking to Your Product Under PED 2014/68/EU
Placement and Documentation for CE Marking
Manufacturers must follow a precise ce marking procedure when certifying a product under ped 2014/68/eu. The pressure equipment directive 2014/68/eu requires that the ce marking appears visibly, legibly, and indelibly on each product. This marking signals that the product meets all essential safety and regulatory standards for pressure equipment in the eu. Companies must also prepare and maintain documentation that supports the ce marking.
The following table summarizes the official requirements for ce marking under the directive:
| Requirement | Description |
|---|---|
| Directive | 2014/68/EU - Pressure Equipment Directive |
| Applicable From | 19 July 2016 |
| Guidance Documents | Guidance on CE marking for professionals, Pressure equipment directive: guidelines |
Manufacturers must attach the ce marking to the product and ensure that all supporting documentation is complete. The technical documentation and the eu declaration of conformity must accompany the product.
| Documentation Type | Description |
|---|---|
| Technical Documentation | Compilation or verification of the Technical Documentation containing descriptions, instruction manual and the performed risk assessments. |
| EU Declaration of Conformity | Verify certificates and draw up the EU Declaration of Conformity affirming compliance with CE product requirements. |
Post-Market Obligations in the EU
After placing the ce marking on a product, manufacturers must fulfill several post-market obligations in the eu. They must monitor the product’s performance and address any safety concerns related to pressure equipment. Companies must keep records of certification and technical documentation for inspection by authorities.
Manufacturers must also update documentation if the product design changes or if new standards apply. They must inform distributors and users about corrective actions when necessary. Regular reviews and updates help maintain compliance with ped 2014/68/eu and support safe use of pressure equipment in the eu market.
Tip: Consistent recordkeeping and proactive communication help manufacturers avoid compliance issues and ensure the continued safety of their products.
Practical Tips and Common Pitfalls in PED 2014/68/EU Certification
Avoiding Delays and Non-Compliance
Manufacturers often encounter challenges during the certification process for pressure equipment under the pressure equipment directive 2014/68/eu. Delays and non-compliance can result from incomplete documentation, inconsistent clinical data, or misunderstanding regulatory requirements. The following table highlights common pitfalls faced by manufacturers of high- and medium-risk products:
| Challenge Description | High-Risk Device Respondents (%) | Medium-Risk Device Respondents (%) |
|---|---|---|
| Invested in clinical evaluation training | 42% | 10% |
| Experienced inconsistencies in clinical data accepted | 46% | 40% |
| Intention to remove devices from the market | 54% | 0% |
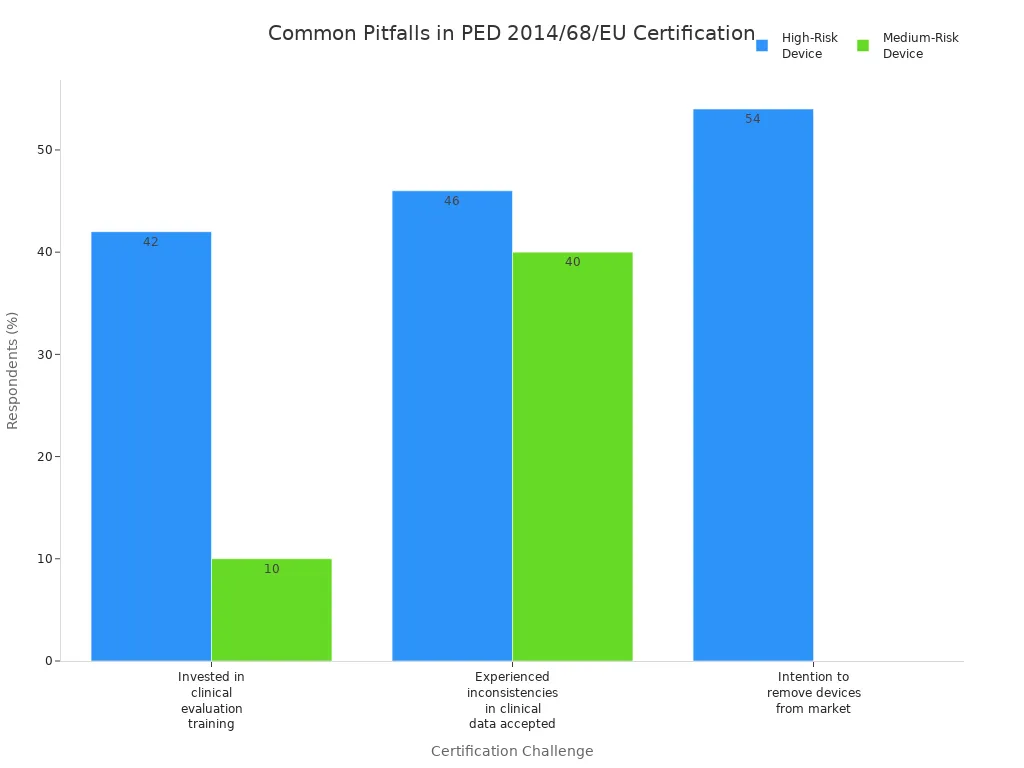
Manufacturers should focus on generating sufficient clinical evidence, understanding the requirements of the MDR, and collaborating with notified bodies. They must prepare comprehensive technical documentation and conduct thorough risk assessments for each product. Regular updates and audits help maintain compliance and avoid costly delays.
Tip: Keep a checklist of all directives and standards. This practice ensures that every requirement is addressed during the ce marking procedure and certification.
Useful Resources and Checklists for Pressure Equipment Directive 2014/68/EU
Access to official resources and structured checklists streamlines the certification process for pressure equipment. The following table outlines essential steps for manufacturers seeking compliance with ped 2014/68/eu:
| Step | Description |
|---|---|
| 1 | Conduct a thorough gap analysis to understand current compliance status. |
| 2 | Develop and implement PED-specific procedures within the QMS. |
| 3 | Train relevant staff and conduct internal pre-audits. |
| 4 | Establish a system for permanent material traceability. |
| 5 | Prepare a Technical File with necessary documentation for the Notified Body. |
Manufacturers should monitor changes in eu regulations and update technical files for each product. Collaboration with notified bodies and adherence to the ce marking procedure support successful certification and market access for pressure equipment.
Manufacturers achieve PED 2014/68/EU certification for pressure equipment by following these essential steps:
- Classify the product under the pressure equipment directive 2014/68/eu.
- Complete the conformity assessment procedure and required testing.
- Prepare and maintain technical files for certification.
- Sign and retain the CE Declaration of Conformity.
- Finish the ce marking procedure before entering the EU market.
Thorough preparation and accurate documentation support successful certification. Companies should seek expert advice for complex products or challenging cases.
FAQ
What products from SHPHE require PED 2014/68/EU certification?
Plate heat exchangers and other pressure equipment from SHPHE require PED 2014/68/EU certification if they operate above 0.5 bar and fall within the directive’s scope.
How long does the PED 2014/68/EU certification process usually take?
The certification process duration depends on product complexity and documentation readiness. Most manufacturers complete it within several weeks to a few months.
Who can help with PED 2014/68/EU certification for plate heat exchangers?
Notified Bodies in the EU provide guidance and assessment services. Manufacturers often consult with industry experts to ensure compliance and streamline the certification process.