What's The Difference Between New And Used Tablet Presses?
Choosing a tablet press involves a fundamental trade-off. New machines offer guaranteed performance and modern technology. Used presses provide a lower initial investment but carry potential risks. The global tablet press machine market is expanding steadily, with projections showing a compound annual growth rate of nearly 5%. This growth highlights the increasing demand for efficient production.
A new press simplifies regulatory adherence, while a lower rotary tablet press machine price for a used unit often comes with challenges in documentation and long-term reliability.
Cost Analysis: Upfront Savings vs. Total Cost of Ownership
The financial evaluation of a tablet press extends far beyond its sticker price. A new machine requires a significant capital outlay, while a used one offers immediate savings. However, a comprehensive analysis of the Total Cost of Ownership (TCO) often reveals a more complex financial picture. Businesses must weigh the upfront cost against long-term operational expenses to make a sound investment.
The Rotary Tablet Press Machine Price for a New Unit
The rotary tablet press machine price for a new unit varies widely. Factors like production capacity, automation features, and manufacturer reputation heavily influence the final cost. Entry-level models provide accessible options for smaller operations, while high-speed, high-output machines represent a more substantial investment for large-scale manufacturing.
The following table and chart illustrate the typical price range for several new rotary tablet press models:
| Model Name | Price (US$) |
|---|---|
| RTP 33 Rotary Pellet Press | 14,530.00 |
| RTP 118 Rotary Tablet Press | 17,110.00 |
| RTP 41 Rotary Tablet Press | 29,010.00 |
| RTP 10i Rotary Tablet Press | 34,390.00 |
| HSTP High Speed Tablet Press Range | 107,370.00 |
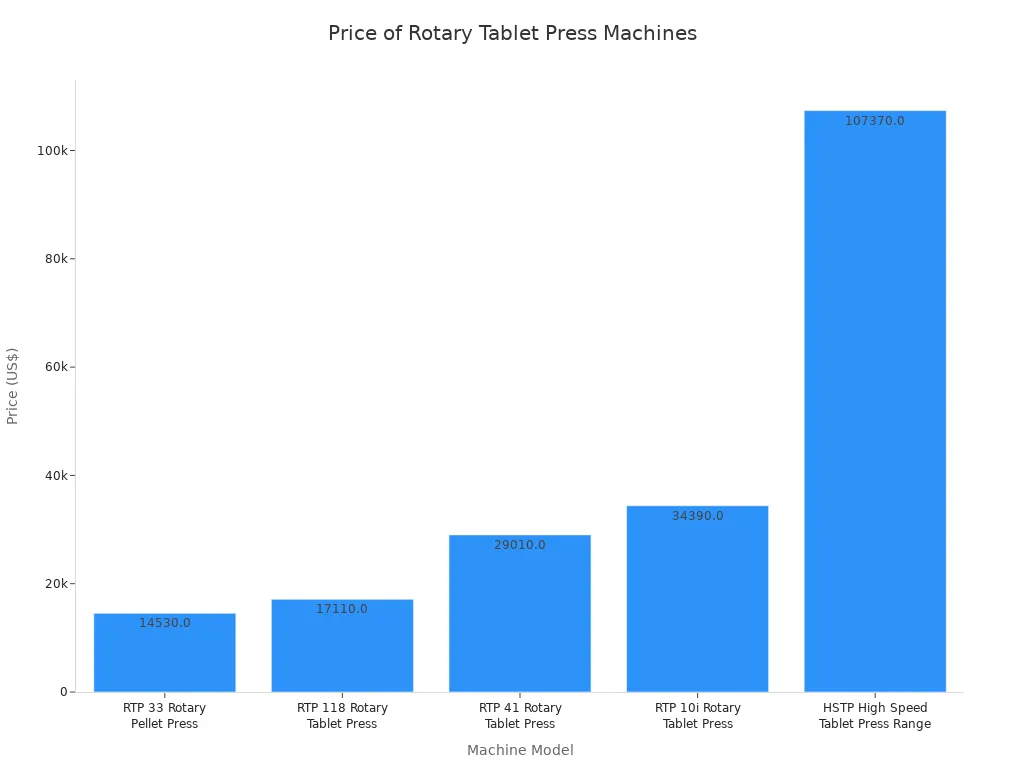
High-speed and multi-layer presses carry a higher initial cost. However, their advanced automation and massive throughput can significantly lower the cost-per-tablet during mass production. This makes the higher rotary tablet press machine price a strategic investment for companies focused on volume. The relationship between production capacity and price is clear, as higher output capabilities demand a larger initial investment.
Calculating the True Cost of a Used Machine
A used machine's low initial purchase price is its main attraction. This can make an attractive rotary tablet press machine price seem like an easy decision. However, this upfront saving can be quickly offset by numerous hidden costs that are not immediately apparent. Calculating the true cost requires a detailed projection of all potential expenses over the machine's expected lifespan.
Note: A thorough evaluation of a used press is critical. Buyers should budget not just for the purchase but for the entire lifecycle, including potential upgrades, repairs, and validation efforts.
Consider these potential additional costs when evaluating a used tablet press:
- Refurbishment and Repairs: Older machines may require immediate replacement of worn components like motors, belts, or electronic controls.
- Tooling and Dies: The existing tooling may be worn, damaged, or unsuitable for the intended product, necessitating a full replacement.
- Validation and Qualification: Bringing a used machine into cGMP compliance can involve significant time and expense for documentation, testing, and validation services.
- Installation and Training: The machine may lack modern user interfaces, requiring specialized training for operators and maintenance staff.
- Downtime: Unexpected breakdowns are more likely with older equipment, leading to costly production delays and lost revenue.
Ultimately, the lower initial rotary tablet press machine price of a used unit must be carefully weighed against these potential long-term expenditures to determine its true value.
Performance and Technology: Modern Efficiency vs. Proven Mechanics

The performance gap between a new and a used tablet press is often defined by technology. New machines integrate advanced mechatronics and software for peak efficiency. Used machines rely on established mechanical systems that are functional but may lack modern optimizations. This technological divide directly impacts production speed, product quality, and operational costs.
New Press: Peak Performance with Advanced Features
A new tablet press represents the pinnacle of manufacturing technology. It delivers superior performance through a combination of precision engineering, automation, and intelligent control systems. These machines are designed for maximum output, minimal waste, and consistent quality, giving businesses a significant competitive edge.
Modern electric tablet presses offer significant energy savings compared to older hydraulic systems. This efficiency can result in an average saving of $1,200 per year. The shift to energy-efficient electric systems also reduces overall operational costs by 15–30%. Furthermore, the precision offered by servo-motors in modern presses can reduce energy waste by another 20%, lowering the total cost of ownership over the machine's lifespan.
Tip: Advanced mechatronic integration, featuring touch screens and real-time data displays, gives operators precise control over production variables like speed and output, while also providing instant fault diagnostics.
Technological advancements in new presses directly contribute to higher production yields and reduced product loss. Key features include:
- Consistent Weight and Hardness Control: Sophisticated sensors and feedback loops make real-time adjustments. This process minimizes product rejects and optimizes overall yield.
- High-Speed Production: Modern presses can produce millions of tablets per hour without sacrificing quality, a vital capability for high-volume manufacturing.
- Computer-Controlled Start-Up: This feature automatically rejects a set number of tablets as the machine ramps up to speed. It prevents out-of-spec "transitional" tablets from entering the good product stream.
- Advanced Extraction Systems: Automatic 'damping' valves precisely modulate the vacuum at turret extraction ports. This optimizes material handling and reduces powder waste.
- Forced Feeding Systems: These systems ensure accurate and uniform filling of powder into the dies. This guarantees consistent active ingredient content in every tablet.
These features work together to create a highly efficient and reliable production environment, ensuring that every batch meets stringent quality standards.
Used Press: Functionality with Potential Limitations
A used tablet press can be a reliable workhorse, built with durable mechanics designed to last for decades. For many applications, its proven functionality is more than sufficient. However, buyers must recognize the inherent limitations that come with older technology. The lower rotary tablet press machine price often reflects a trade-off in performance, efficiency, and automation.
While mechanically sound, an older press is more susceptible to specific types of wear and tear. A thorough inspection is critical to identify potential issues before purchase. Common points of failure in older models include:
- Feeding Mechanism Malfunctions: This can lead to uneven powder distribution and inconsistent tablet weights.
- Die and Punch Wear: Worn tooling directly affects tablet shape, quality, and consistency.
- Ejection Problems: Poor lubrication or worn components can cause tablets to break or chip during ejection.
- Material Adhesion: Powder sticking to punch faces results in rough or deformed tablets.
- General Wear: Neglected cleaning and maintenance can lead to residue buildup, which contaminates batches and affects overall performance.
These machines typically lack the advanced data monitoring, energy-efficient servo-motors, and automated quality control systems found in new models. This absence can lead to higher energy consumption, increased reliance on manual oversight, and a greater potential for production inconsistencies.
Key Features Driving Modern Tablet Production

Modern tablet presses are defined by a suite of advanced features that boost efficiency, ensure compliance, and protect operators. These innovations move beyond basic mechanics, integrating intelligent systems that deliver precision and reliability. The result is a streamlined production process from start to finish.
Advanced Control and Mechatronics
New tablet presses combine mechanical and electronic engineering (mechatronics) to achieve unparalleled control. Digital systems with touch screens provide operators with real-time data on speed, output, and potential faults. This integration allows for precise, automated adjustments that maintain quality and reduce manual oversight.
A key advantage of modern mechatronics is the use of product recipes. These systems store optimized parameters for press speed, dosing, and force. Operators can recall a recipe to automatically configure the machine, ensuring the product is within specification from the start with only minor adjustments needed.
These systems use feedback loops to dynamically manage production. Integrated sensors continuously track key parameters like compression force and tablet weight. If the system detects a deviation, it automatically adjusts to maintain consistency, minimizing waste and ensuring uniform active ingredient content in every tablet.
GMP-Compliant Design and Dust Collection
Adherence to Good Manufacturing Practices (GMP) is non-negotiable, and modern presses are built with compliance in mind. They often feature a fully enclosed stainless steel shell that prevents cross-contamination and simplifies cleaning. A transparent protective cover allows for clear observation of the machine's operation while keeping the compression zone contained.
A critical component of GMP-compliant design is an effective dust collection system.
- Integrated dust extraction ports are placed at key points of dust generation.
- These ports connect to high-efficiency collectors that absorb airborne powder.
- This process keeps the pressing chamber clean, prevents cross-contamination between batches, and protects operators from inhaling harmful particles.
Designs that separate the processing and mechanical areas further simplify cleaning and maintenance, reducing downtime between production runs.
Versatility and Safety Mechanisms
Modern tablet presses offer significant versatility. Many models feature interchangeable dies, allowing them to produce a wide variety of tablet sizes and shapes, from standard round tablets to complex, irregular designs. This flexibility enables manufacturers to adapt to diverse product requirements with a single machine.
Safety is a paramount concern, and new presses incorporate multiple protective features.
- Emergency Stop Devices: These allow operators to halt all machine functions immediately in a hazardous situation.
- Overload Protection: This system automatically stops the press if compression force exceeds safe limits, preventing costly damage to tooling and machine components.
- Interlocked Guards: Protective covers are fitted with sensors that stop the machine if a guard is opened during operation, preventing access to moving parts.
These integrated safety mechanisms work together to protect both the equipment and the personnel operating it.
Regulatory Compliance and Validation: The cGMP Hurdle
Navigating the regulatory landscape is a critical challenge in pharmaceutical manufacturing. The U.S. FDA’s Current Good Manufacturing Practice (cGMP) regulations, detailed in 21 CFR Parts 210 and 211, set strict standards for equipment. These rules require that every machine's functionality is clearly defined, validated, and documented. A new press simplifies this process, while a used one can present significant hurdles.
New Press: Built-in Compliance and Documentation
Manufacturers design new tablet presses to meet cGMP standards from the ground up. They provide a comprehensive validation package that streamlines the qualification process. This documentation bundle typically includes:
- Installation Qualification (IQ): Verifies the machine is installed correctly.
- Operational Qualification (OQ): Confirms the machine operates according to its specifications.
- Performance Qualification (PQ): Demonstrates the press consistently produces quality tablets under real production conditions.
Modern press software also adheres to 21 CFR Part 11 for electronic records and signatures. Features like secure audit trails, limited system access, and unique user identification ensure data integrity and traceability.
This built-in compliance and thorough documentation package significantly reduces the time and resources needed to get a new machine operational in a regulated environment.
Used Press: The Validation and Documentation Challenge
A used tablet press places the full burden of validation on the buyer. These machines often arrive with significant documentation gaps, such as incomplete validation records or a lack of historical data. The buyer must retroactively prove the machine meets cGMP standards, a task that can be both costly and time-consuming.
Common challenges include the absence of detailed risk assessments and poorly defined validation protocols. This lack of information makes it difficult to qualify the equipment for its intended use.
Note: The FDA reports that improper equipment design and maintenance account for 15% of all cGMP-related warnings. A used machine with an unknown history poses a direct risk of non-compliance.
Without the original manufacturer's support, creating the necessary IQ, OQ, and PQ documentation becomes a complex project. This validation challenge can delay production and introduce significant regulatory risk, potentially negating the initial cost savings.
Support, Warranty, and Parts Availability
The long-term value of a tablet press depends heavily on the support structure behind it. A new machine provides a safety net of guarantees and expert help. A used machine often leaves the buyer to navigate maintenance and repairs alone.
New Press: Comprehensive Manufacturer Guarantee
Purchasing a new tablet press provides buyers with a robust manufacturer guarantee. This package offers peace of mind and protects the investment. Most manufacturers provide a standard warranty that covers the machine for a set period.
- Many companies, including Hunan Grand Packing Machinery and LFA Machines, offer a one-year warranty from the date of purchase.
- This guarantee typically includes free spare parts for any machine quality issues.
- Some suppliers even include a set of wear and tear parts with the initial shipment.
Beyond the warranty, manufacturers deliver a full suite of support services. These programs ensure operators can maximize the machine's potential and resolve issues quickly.
Support packages often include expert technical assistance, remote troubleshooting, and software updates. Comprehensive training programs also equip teams with the skills for effective operation and maintenance, ensuring long-term performance and reliability.
Used Press: "As-Is" Purchases and Sourcing Challenges
Used tablet presses are almost always sold "as-is," a term that carries significant risk. This means the buyer accepts the equipment in its current condition without any guarantees from the seller.
The seller makes no warranties regarding the equipment's performance or fitness for a particular purpose. The buyer assumes all risks and is not protected from losses, damages, or liabilities resulting from equipment failure. It becomes the buyer's sole responsibility to test the machine and ensure it meets their needs.
Sourcing replacement parts presents another major challenge. While some specialized suppliers stock over 250,000 parts for older brands like Colton and Stokes, finding the right component is not always simple. Locating a specific part for an obsolete model can lead to extended downtime and production delays. This sourcing uncertainty adds another layer of risk to the lower upfront rotary tablet press machine price.
A new rotary tablet press is the optimal choice for businesses prioritizing cGMP compliance, maximum efficiency, and long-term reliability. A used press can be a strategic investment for companies with strong in-house technical teams and a primary focus on minimizing initial capital expenditure.
Your final decision should balance budget, production needs, technical capabilities, and risk tolerance. A thorough evaluation of these factors will guide you to the right equipment for your operation.
FAQ
What is the most critical factor when considering a used tablet press?
A buyer's in-house technical capability is the most important factor. A strong maintenance team is essential. They must handle potential repairs, source parts, and manage the validation process without extensive manufacturer support, which is a common scenario with used equipment.
How do new tablet presses simplify GMP compliance?
New presses are designed to meet GMP standards. Manufacturers provide a full validation package, including IQ, OQ, and PQ documentation. Their modern software often includes features like secure audit trails, which helps ensure data integrity and regulatory adherence from the start.
Can a modern tablet press produce different tablet shapes?
Yes, modern presses offer excellent versatility. Many models feature interchangeable dies. This design allows a single machine to produce tablets of various sizes and shapes, including standard round tablets and complex, irregular designs, adapting to diverse product requirements.
What safety features are standard on new tablet presses?
New machines integrate multiple safety mechanisms to protect operators and equipment. Key features include emergency stop devices for immediate shutdown, overload protection to prevent tooling damage, and interlocked guards that stop the machine if a protective cover is opened during operation.
See Also
Essential Guide: Selecting the Best Tablet Sampling Machines for Your Lab
Defining New Energy Vehicles: Key Characteristics and Future Transportation
Smart Savings: Understanding Pressure Plate Essentials for Your Wallet
Boosting Pharmaceutical Efficiency: Modern Capsule Decapsulation Technology Explained
Top Capsule Checkweighers: A Buyer's Guide for 2025 Considerations