Electrochemical and Ozone The New Era of Sewage Treatment

More than 80% of the world’s sewage is discharged into the environment untreated.
New electrochemical and ozone methods offer efficient, chemical-free solutions for this global challenge. These technologies effectively neutralize persistent pollutants that traditional sewage treatment systems often miss. They represent a sustainable future for clean water by reducing sludge and harmful byproducts.
Key Takeaways
- New water treatments use electricity and ozone. They clean water better than old ways.
- These new methods break down bad stuff in water. They make fewer harmful chemicals.
- Combining these treatments makes water even cleaner. It helps get useful things from wastewater.
What is Electrochemical Treatment?
Electrochemical treatment uses electricity to clean water. This modern approach applies electrical energy to drive chemical reactions. These reactions can either convert pollutants into harmless substances or separate them from the water. It is a powerful tool in the new era of sewage treatment. The main methods include electrocoagulation, electrooxidation, and electroflotation.
The Science of Electrocoagulation
Electrocoagulation is a process that removes contaminants from water using an electric current. It works by destabilizing pollutants that are suspended in the water. The system uses metal plates, called electrodes, which are submerged in the wastewater. When an electric current is applied, the positive electrode (anode) dissolves. It releases metal ions, like aluminum or iron, into the water.
These positively charged ions neutralize the negative charges of pollutants like suspended solids and heavy metals. Without their charge, the pollutants no longer repel each other. They begin to clump together, forming larger particles called flocs. These flocs are heavy and can be easily removed from the water through sedimentation or filtration.
How do the electrodes react? The chemical reactions at the electrodes are the engine of the process. Using an aluminum anode as an example, the reactions are:
| Electrode | Reaction |
|---|---|
| Anode | Al → Al³⁺(aq) + 3e⁻ |
| Cathode | 3H₂O + 3e⁻ → 3/2H₂ + 3OH⁻ |
This in-situ generation of coagulants is highly efficient for removing charged particles and metals from wastewater.
How Electrooxidation Breaks Down Contaminants
Electrooxidation (EO) is a powerful process that destroys stubborn organic pollutants. It breaks them down into simple, harmless molecules like carbon dioxide and water. This is achieved through two primary mechanisms:
- Direct Oxidation: Pollutants are destroyed directly on the surface of the anode. They transfer electrons to the electrode and are broken apart in the process.
- Indirect Oxidation: The process generates powerful cleaning agents, called radicals, within the water itself. These highly reactive species, such as hydroxyl radicals (•OH) and sulfate radicals (SO₄•⁻), hunt down and destroy organic pollutants.
Sulfate radicals are especially effective. They have a higher redox potential than hydroxyl radicals over a wide pH range. This means they react faster and more aggressively with tough contaminants. This strong oxidative power allows EO to achieve remarkable results. For example, studies have shown that electrooxidation can achieve greater than 99% removal efficiency for certain pesticides.
Key Benefits Over Conventional Methods
Electrochemical methods offer significant advantages over traditional water treatment systems. They are often more efficient, cost-effective, and environmentally friendly.
1. Lower Operating Costs While the process requires electricity and replacement electrodes, the overall operating costs are often low. There is less need for transporting and storing bulk chemicals. The total cost can be estimated with a simple formula.
The operating cost is a sum of energy, electrode, and chemical costs:
Operating Cost = aC_energy + bC_electrode + DWhereais the electricity price,bis the electrode price, andDis the cost of any other chemicals.
2. Superior Energy Efficiency Some advanced electrochemical systems can even produce more energy than they consume. When combined with processes like anaerobic digestion, the energy generated from biogas can exceed the electrical energy needed for treatment. This creates a net positive energy balance.
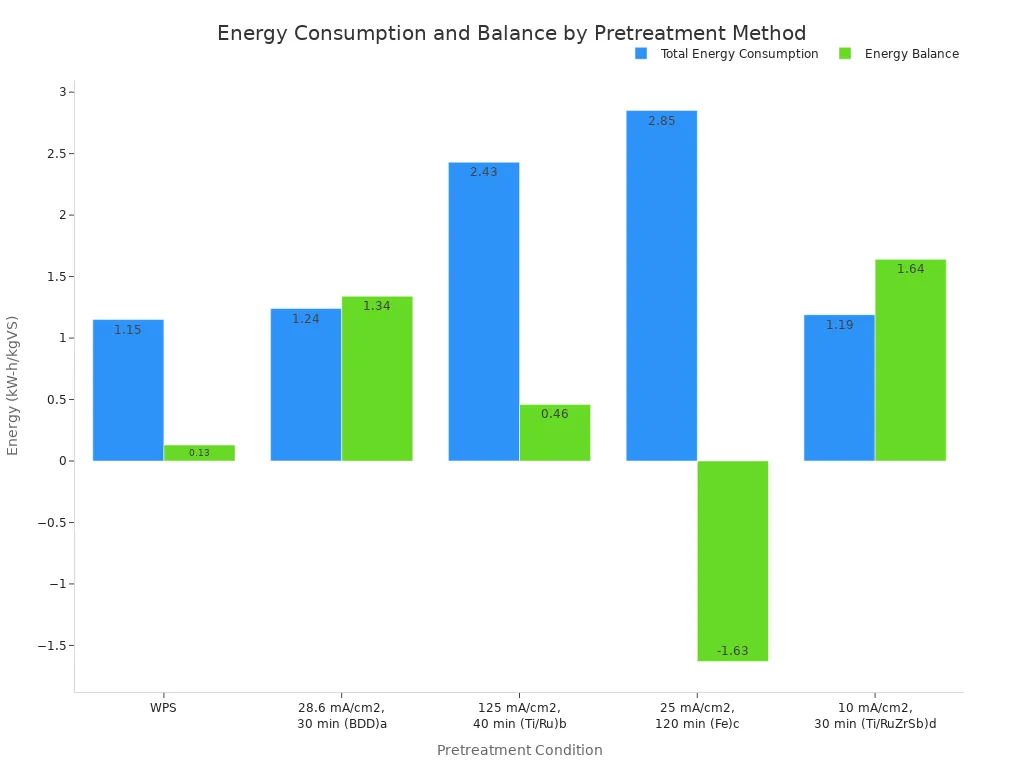
As the chart shows, one electrochemical system (Ti/RuZrSb) had a net positive energy gain of 1.64 kW-h/kgVS, making it a highly sustainable option.
3. Reduced Sludge Production Traditional methods produce large volumes of wet sludge, which is costly to handle and dispose of. Electrochemical techniques like electro-dewatering can dramatically reduce the moisture content of sludge. This makes the final solid waste much lighter and more compact, significantly cutting down on disposal costs.
How Ozone Treatment Transforms Water Purification
Ozone treatment is a powerful water purification method that uses ozone gas (O₃) to clean water. Ozone is a molecule made of three oxygen atoms. It is a strong oxidizing agent, meaning it can effectively break down and remove a wide range of contaminants. This technology is used in large-scale industrial and municipal water systems to provide safe, clean water.
Defining Ozonation in Water Treatment
Ozonation is the process of adding ozone to water to achieve disinfection and purification. The ozone itself is created on-site, so there is no need to transport or store dangerous chemicals. The generation process is quite simple:
- An ozone generator takes in regular oxygen (O₂).
- A high-voltage electrical charge, similar to lightning in a thunderstorm, is applied to the oxygen. This is called a corona discharge.
- The electrical charge splits the oxygen molecules into single oxygen atoms.
- These single atoms quickly combine with other oxygen molecules (O₂) to form ozone (O₃).
This freshly made ozone gas is then bubbled into the water, where it immediately starts working. Its applications are incredibly diverse, making it a versatile tool for water quality management.
Common Uses of Ozone Treatment 💧
- Municipal Water: It disinfects drinking water, removes unpleasant tastes and odors, and reduces iron and manganese. It is also used in wastewater plants to disinfect the final water before it is returned to the environment.
- Industrial Wastewater: Ozone removes tough pollutants like phenols, cyanide, and color from industrial discharge. It also helps keep sumps and tanks clean and free of odors.
- Food & Beverage: It is used to disinfect bottled water and to wash fruits and vegetables, killing harmful bacteria like E. coli.
- Aquaculture: In fish farming, ozone improves water quality by removing waste products and disinfecting the water.
While effective, setting up an ozone system involves initial costs. For a large-scale plant, the ozone generator is the main expense. For smaller operations, the costs can be broken down.
| Cost Category | Example Startup Amount |
|---|---|
| Equipment Purchase (Generators) | $25,000 |
| Service Vehicles | $70,000 |
| Tools & Safety Gear | $7,000 |
| Total Major Capital | $102,000 |
Ongoing operational costs are mainly for electricity to run the generators and for staff salaries.
The Mechanism of Ozone Disinfection
Ozone is a powerful disinfectant because of its high oxidation potential. It physically destroys pathogens rather than just poisoning them. The process works by breaking down the cell walls of microorganisms like bacteria and viruses.
Here is how it happens:
- Ozone (O₃) is unstable and quickly breaks down in water, releasing a highly reactive single oxygen atom.
- This oxygen atom attacks the protective outer wall of a microbe.
- The attack creates a hole in the cell wall, causing the cell to leak its contents.
- With its wall breached, the microbe can no longer survive and is destroyed.
This process, known as cell lysis, is extremely fast and effective. Ozone is particularly good at destroying Gram-negative bacteria, which have thinner cell walls. Gram-positive bacteria have thicker walls, making them slightly more resistant, but ozone can still effectively neutralize them.
Advantages Over Traditional Chlorination
Ozone offers several key advantages over chlorination, the most common method of disinfection in sewage treatment. These benefits make it a safer and more effective choice for modern water purification.
1. Faster and Stronger Disinfection Ozone works much faster than chlorine. It can neutralize bacteria and viruses in seconds, whereas chlorine can take many minutes. In fact, ozone is estimated to be 3,000 times faster than chlorine at disinfection. This superior efficiency is measured by the CT value (Concentration x Time), which shows how much disinfectant and contact time are needed to kill a certain number of microbes. A lower CT value is better.
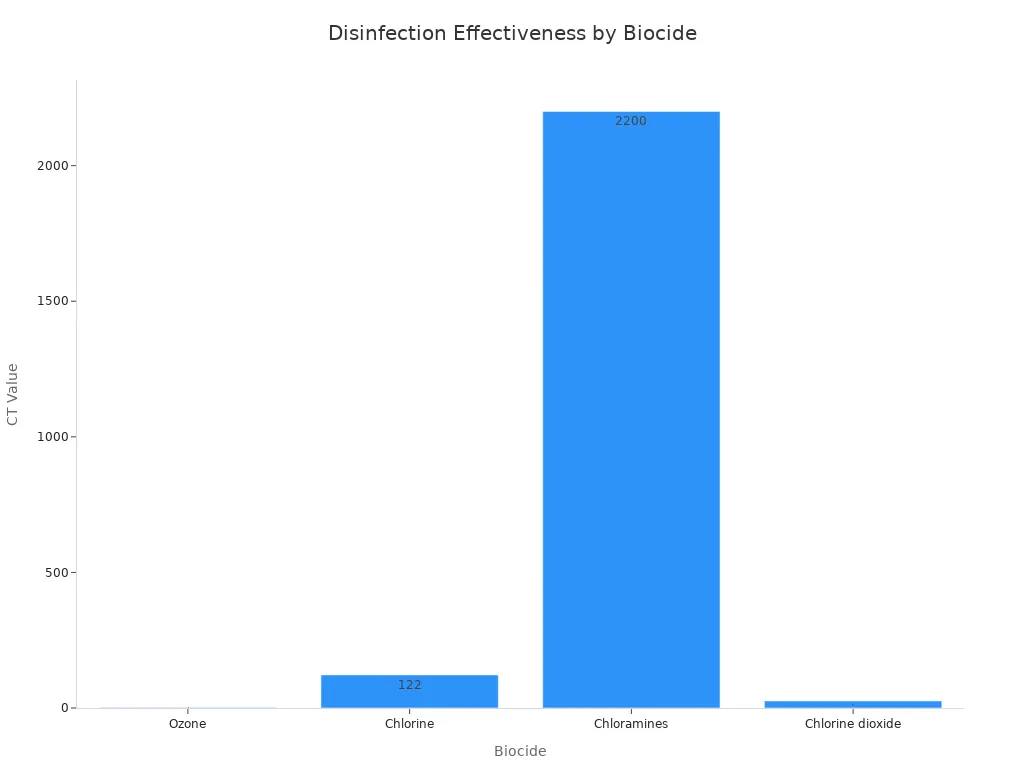
As the chart shows, ozone's CT value is dramatically lower than chlorine's, proving it gets the job done more efficiently.
2. Effective Against Chlorine-Resistant Pathogens Some dangerous pathogens are not affected by the standard doses of chlorine used in water treatment. Cryptosporidium and Giardia are two such protozoa that can cause serious illness. Ozone, however, is extremely effective at inactivating them. It breaks through their tough outer shells, something chlorine struggles to do. This makes ozonation a critical tool for ensuring water is safe from a wider range of microorganisms.
3. Fewer Harmful Byproducts When chlorine reacts with organic matter in water, it can form harmful chemicals called disinfection byproducts (DBPs), such as trihalomethanes (THMs). Some of these DBPs are linked to health risks.
Using ozone before chlorination can significantly reduce the formation of these dangerous compounds. Studies have shown that this combined approach can:
- Lower DBP formation by over 28%.
- Reduce substances that cause mutations in water.
- Result in cleaner, safer drinking water with fewer unknown chemical byproducts.
By breaking down organic matter first, ozone leaves less "fuel" for chlorine to react with, leading to a healthier final product.
The Combined Power: Advanced Oxidation Processes
When electrochemical and ozone treatments are used together, they create a super-powered cleaning system called an Advanced Oxidation Process (AOP). This approach is more effective than using either method alone. It generates extremely reactive molecules that destroy pollutants at a remarkable speed.
Synergies of Electrochemical and Ozone Methods
The magic behind AOPs is their ability to create hydroxyl radicals (•OH). These are some of the most powerful oxidizing agents known. They are formed right inside the water, ready to attack contaminants. The combination of ozone and electricity enhances this process.
How are Hydroxyl Radicals Made? 🧪 AOPs use energy and other chemicals to produce these radicals in several ways:
- Ozone-based: Ozone reacts with ions in the water to start a chain reaction that produces hydroxyl radicals.
- UV Light: Shining UV light on hydrogen peroxide (H₂O₂) or hypochlorous acid (HOCl) splits them into hydroxyl radicals.
- Fenton Process: A reaction between iron and hydrogen peroxide also creates these powerful cleaning agents.
This synergy makes the overall sewage treatment process much more powerful.
Boosting Treatment Speed and Efficiency
Combining methods dramatically improves performance. An integrated electrochemical-ozone system removes far more contaminants than ozone can by itself. This boost in efficiency means cleaner water is produced faster. The data clearly shows this improvement.
| Method | COD Reduction (%) | BOD₅ Reduction (%) |
|---|---|---|
| Ozone treatment alone | 60 | 60 |
| Integrated electrochemical–ozone | 84 | 79 |
This combined approach achieves better results and works best at a neutral pH of 7, making it more practical for many facilities.
Targeting and Eliminating Tough Pollutants
AOPs are experts at destroying stubborn pollutants that other systems miss. The combination of ozonation and electro-oxidation (O₃-EO) is especially good at breaking down industrial chemicals like phenol. Studies show this hybrid method can:
- Reduce the treatment time for phenol by half.
- Completely eliminate the water's toxicity.
- Prevent buildup on the electrodes, helping the system run smoothly.
By working together, these technologies provide a robust solution for eliminating even the most challenging contaminants from water.
Innovations in Modern Sewage Treatment

Modern innovations are making advanced sewage treatment more accessible. These new technologies can be added to existing facilities or used to create new, smaller systems. They offer flexible solutions for cities, industries, and even remote villages.
Upgrading Municipal Wastewater Plants
Cities can upgrade their current wastewater plants with electrochemical and ozone systems. These technologies can be added as a final polishing step. This removes any tough pollutants that older methods miss. It improves the plant's overall performance without needing a complete and costly rebuild. This approach helps municipalities meet stricter environmental rules and produce cleaner water.
Case Studies in Industrial Effluent
Industrial wastewater often contains complex chemicals that are hard to remove. Advanced oxidation methods provide a powerful solution. For example, Boromond has used electro-oxidation to treat wastewater from pharmaceutical and textile factories. The process removes active drug ingredients and stubborn dyes. Another study treated textile wastewater in Tunisia using solar-powered Electro-coagulation and Electro-Fenton processes. This approach achieved 100% color removal and degraded up to 97% of the Chemical Oxygen Demand (COD).
Decentralized Treatment for Remote Areas
Electrochemical and ozone systems are perfect for remote areas without a central water grid. These small, self-contained units can provide safe drinking water for entire communities.
- They create disinfectants directly from the water, so no dangerous chemicals need to be stored.
- The systems require very little energy and can run on a single solar panel.
- They are easy to scale up or down and require minimal maintenance.
- Methods like electrochlorination provide a safe, controlled way to disinfect water.
The Future Outlook for Clean Water

The future of clean water is not just about purification. It is also about recovering valuable resources from what we once called waste. Electrochemical and ozone technologies are leading this change, creating a more sustainable and circular water system.
Enabling Resource Recovery from Wastewater
Wastewater is full of useful materials. Modern electrochemical methods can pull these resources directly from the water. These systems can simultaneously recover phosphorus, nitrogen, and heavy metals. This turns a costly disposal problem into a source of valuable products. The technology is so advanced it can even capture different forms of phosphorus, a key ingredient in fertilizer. This process helps us reuse important nutrients instead of letting them pollute our environment.
The Role in a Circular Water Economy
These advanced treatments are essential for a circular water economy, where nothing is wasted. They help us achieve this goal in several ways:
- Creating Energy: Biogas can be captured during treatment. This gas can then power the plant itself, reducing energy costs.
- Reusing Water: Purified water can be used for farming and irrigation. This is especially helpful in areas with water shortages.
- Recycling Nutrients: Recovered phosphorus can be used to enrich soil. This supports sustainable farming and reduces the need for mining new resources.
By turning wastewater into a resource, these technologies reduce waste and create value. This is the core principle of a circular economy.
Overcoming Challenges to Widespread Adoption
Despite their benefits, these technologies face some hurdles. The biggest challenge is cost.
- High Initial Investment: Ozone generators and advanced sensors are expensive. The total installation cost can range from $50,000 to $500,000, which is a major barrier for smaller companies and developing regions.
- Operating Costs: Technical issues can also increase costs. Electrodes can develop a buildup that reduces their effectiveness, a problem called passivation. These systems can also use a lot of electricity, making them expensive to run.
Overcoming these financial and technical challenges is key to making these powerful water treatment solutions available everywhere.
Electrochemical and ozone treatments are a major step forward in wastewater management. Adopting these methods is vital for a future with cleaner water and a healthier planet. These innovations help build stronger and more efficient water systems for everyone, supporting global goals like the UN's push for clean water and sanitation.
FAQ
Are these new water treatments safe?
Yes, they are very safe. 🌎 These methods use electricity and oxygen to clean water. They produce fewer harmful chemicals than traditional methods, making them much better for the environment.
Why isn't everyone using these methods yet?
The main reason is cost. The equipment for ozone and electrochemical systems is expensive to buy and install. This makes it difficult for smaller towns or developing areas to adopt.