What to Look for in a Rotary Tablet Press Machine
Selecting the right rotary tablet press machine is a critical investment for manufacturers. The global market is expanding, with some reports projecting a Compound Annual Growth Rate (CAGR) of up to 8.5% through 2033, driven by the pharmaceutical sector's growth. A thorough evaluation helps businesses match technical specifications to production needs. Buyers must also verify performance features to ensure product quality and regulatory compliance. Finally, assessing supplier reliability and the total cost of ownership secures long-term value and operational efficiency in a competitive landscape.
Evaluating Production and Technical Specifications

Buyers must first analyze a machine's core specifications. These technical details determine if a press can meet production targets and handle specific formulations. A careful review of these numbers provides a clear foundation for comparing different models.
Production Capacity and Speed
Production capacity, measured in tablets per hour, is a primary consideration. This figure directly relates to the turret's rotation speed and the number of stations. Machine capabilities vary significantly based on their intended scale of operation.
| Scale | Example Machine | Max. Production Capacity/Hour |
|---|---|---|
| Small | Desktop Presses | 1,500 - 4,800 |
| Medium | RTP 33 | 99,000 |
| Large | HSTP 40 | 250,000 |
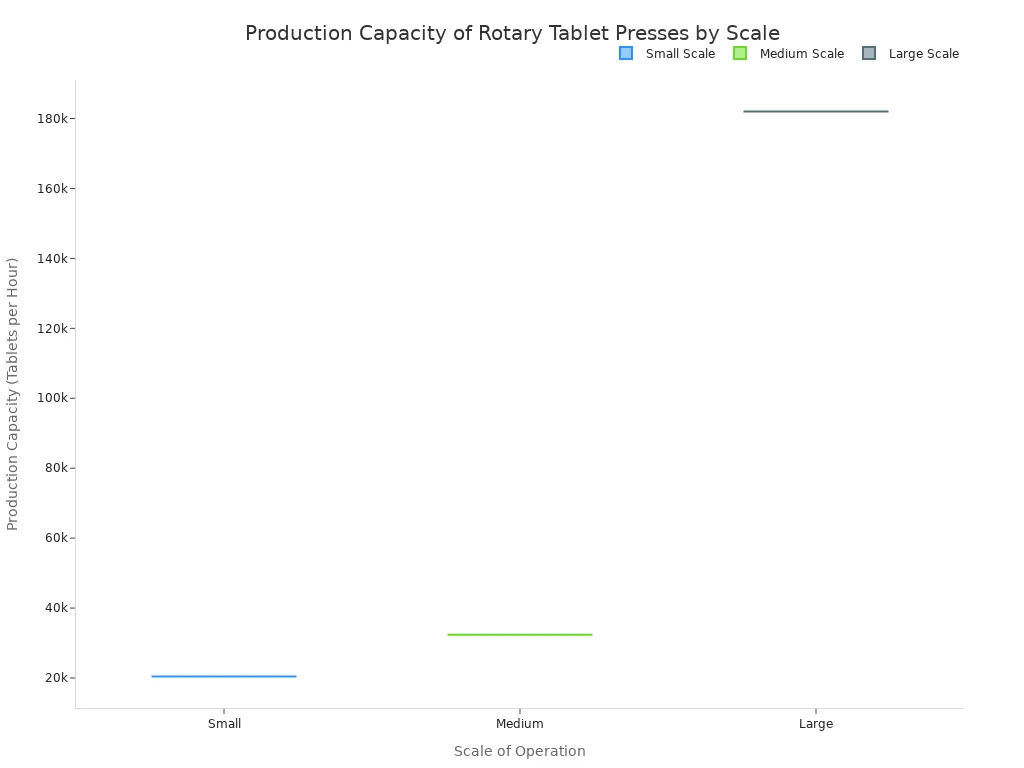
While higher speeds increase output, they can challenge tablet weight and hardness consistency. Advanced machines overcome this issue. They use electronic monitoring and dual compression systems to maintain excellent uniformity even at high throughputs.
Main and Pre-Compression Force (kN)
Compression force, measured in kilonewtons (kN), is essential for forming a durable tablet. The main compression force determines the final hardness and density of the tablet. For example, studies on ibuprofen have used compaction forces ranging from approximately 5 kN to 30 kN.
Pre-compression is a lower-force stage that occurs before main compression. Its primary role is to remove entrapped air from the powder. This de-aeration process helps prevent common tablet defects like capping and lamination.
Pro Tip: Applying the correct pre-compression force is critical. The ejected compact should be consolidated but still crumble easily when squeezed. This indicates proper air removal without premature bonding of particles.
Number of Punch Stations
The number of punch stations on the turret directly correlates with the maximum output of a rotary tablet press machine. Each station holds a die and a set of punches to produce one tablet per turret revolution. Therefore, a press with more stations can produce more tablets per hour at the same rotational speed. Manufacturers often increase the station count to achieve higher production capacities, with modern presses producing over 250,000 tablets per hour.
Maximum Tablet Diameter and Depth of Fill
The maximum tablet diameter and depth of fill are fundamental specifications that define a machine's production capabilities. These two parameters determine the physical size and weight range of the tablets a press can manufacture. Buyers must carefully match these specifications to their current and future product requirements.
Maximum tablet diameter refers to the largest possible width of a tablet. This dimension is directly linked to the type of tooling the machine uses. For instance, a press equipped with 'D' type tooling can produce larger tablets than one using 'B' type tooling. This specification is crucial for companies producing a diverse portfolio, from small pharmaceutical pills to large veterinary or nutraceutical tablets.
Depth of fill controls the volume of powder that fills the die cavity before compression. This adjustable setting is critical for controlling tablet weight and, consequently, the dosage of the active ingredient. Key factors influencing this setting include:
- Tablet Weight: A heavier tablet requires a greater depth of fill.
- Powder Density: Formulations with low bulk density need more volume to achieve the target weight.
- Tablet Thickness: The final desired thickness also influences the required fill depth.
Expert Insight: A machine with a generous maximum tablet diameter and a wide range of fill depths offers greater manufacturing flexibility. It allows a business to adapt to new product formulations or enter different markets without needing to invest in new equipment.
Ultimately, these specifications set the boundaries for product development. A machine must accommodate the largest and heaviest tablet a manufacturer plans to produce. Evaluating these limits ensures the chosen rotary tablet press can handle the full scope of a company's production needs.
Understanding Tooling and Formulation Needs
A rotary tablet press must align with a company's specific tooling and formulation requirements. The machine's compatibility with different tooling types, its turret design, and the materials of its contact parts directly impact production flexibility, efficiency, and product integrity.
Tooling Type and Flexibility (B, D, BB, DB)
Tooling defines the size, shape, and characteristics of the final tablet. The most common standards are B and D, with sub-types like BB and DB offering further options. B-tooling, or American type tooling, has a smaller punch head diameter. It is ideal for producing smaller tablets where size precision is critical, such as in pediatric medications. In contrast, D-tooling, also known as Euro or IPT type tooling, features a larger punch head. This allows for the production of larger tablets and can support higher output rates.
The choice between tooling types depends on the product portfolio.
| Feature | B-Type Tooling | D-Type Tooling |
|---|---|---|
| Primary Use | High-volume, standard tablets | Specialized or large tablets |
| Tablet Design | Conventional flat or convex faces | Unique shapes, scores, or embossing |
| Punch Size | Smaller diameter | Larger diameter |
Turret Interchangeability
Modern tablet presses often feature an interchangeable turret design. This capability allows manufacturers to swap out the entire turret, which holds the punches and dies. An interchangeable system provides significant operational advantages.
- Increased Flexibility: It enables the use of different tooling types (e.g., B and D) on a single machine.
- Faster Changeovers: Operators can prepare a second turret with pre-installed tooling, drastically reducing downtime between production runs.
- Higher Productivity: Quick, safe turret removal and easy-to-clean surfaces minimize the time spent on cleaning and setup, maximizing production uptime.
Material and Formulation Compatibility
The materials used for machine parts that contact the powder are critical. These parts must resist corrosion and abrasion to prevent product contamination and ensure longevity. For many pharmaceutical applications, 316L stainless steel is the material of choice. This low-carbon alloy offers excellent corrosion resistance, making it highly effective against acidic or saline formulations. Its durability ensures it can withstand harsh cleaning agents and maintain a hygienic surface, meeting strict GMP standards. Choosing a press with robust, formulation-compatible materials protects both the product and the investment.
Forced Feeding System for Powder Uniformity
Achieving consistent tablet weight is a primary goal in tablet manufacturing. A forced feeding system is an essential component for this task, especially when working with powders that have poor flow characteristics. This system actively manages powder delivery into the die cavities, ensuring each tablet contains the correct amount of formulation.
A forced feeder uses rotating paddles to mechanically push powder into the dies. This active process overcomes the limitations of traditional gravity-fed systems. It breaks up powder clumps and ensures a consistent, complete fill, regardless of the powder's natural flow properties. Gravity-dependent methods, in contrast, are susceptible to weight variations when powders do not flow freely. The mechanical action of a forced feeder is therefore crucial for achieving precise tablet weights with challenging formulations.
The table below highlights the key differences between the two systems:
| Feature | Forced Feeding System | Gravity Feeding System |
|---|---|---|
| Mechanism | Mechanical (rotating paddles/wheels) | Relies on gravity |
| Powder Flow | Overcomes poor flow characteristics | Requires excellent flowability |
| Die Filling | Ensures complete and consistent filling | Prone to variations with inconsistent density |
| Weight Uniformity | Enhanced uniformity, consistent tablet weights | Sensitive to density variations |
| Suitable Powders | Poorly flowing, critical weight precision | Free-flowing, non-sticky |
Key Takeaway: By ensuring accurate filling of powder into the tablet pressing area, a forced feeding system guarantees uniform active ingredient content. This feature is indispensable for high-speed production and for formulations where dosage accuracy is non-negotiable. It directly contributes to higher product quality and reduces the rate of rejected tablets.
Key Performance Features of a Rotary Tablet Press Machine

Modern performance features transform a standard tablet press into a highly efficient and reliable production asset. These systems enhance control, optimize speed, and guarantee product quality. They are essential for meeting the stringent demands of today's manufacturing environment.
Automation and Control Systems (HMI/PLC)
Advanced automation is central to modern tablet production. A Human-Machine Interface (HMI), often a touch screen, provides a user-friendly dashboard for operators. It displays critical data like production speed, output count, and system faults in real time. The HMI works with a Programmable Logic Controller (PLC) to manage machine functions. This mechatronic integration gives operators precise control over the entire tableting process, simplifying operation and improving consistency.
Variable Speed Control
Variable speed control offers significant operational flexibility. It allows manufacturers to fine-tune the machine's speed for different stages of production and for various powder formulations. This precise adjustment is crucial for optimizing tablet quality and efficiency. Key benefits include:
- Formulation Flexibility: Operators can tailor the press speed to match the unique compressibility of different powders.
- Production Efficiency: The ability to start slow for calibration and then ramp up to full speed minimizes waste during setup.
- Reduced Tablet Defects: Optimizing compression dwell time helps prevent common issues like capping and lamination, improving overall yield.
- Enhanced R&D Capabilities: Researchers can quickly test how different speeds affect tablet properties, accelerating formulation development.
Automated Tablet Rejection System
An automated tablet rejection system is vital for quality assurance. This feature uses sophisticated technology to identify and remove defective tablets from the production line automatically. A modern rotary tablet press machine may employ high-speed cameras and advanced AI vision systems. These tools detect physical flaws like cracks, chips, and incorrect shapes. Additionally, accurate checkweighers ensure every tablet meets the precise weight specification, which is critical for dosage control. This automated process guarantees that only compliant products reach the final packaging stage.
Integrated Dedusting and Dust Collection
Effective dust management is a non-negotiable aspect of modern tablet production. An integrated dedusting and dust collection system plays a dual role. It protects product integrity and ensures a safe working environment. These systems are essential for maintaining compliance with Good Manufacturing Practices (GMP).
A deduster removes fine powder residue from the surface of freshly compressed tablets. This step is critical for several reasons:
- Prevents Cross-Contamination: It ensures that loose powder from one batch does not mix with subsequent products.
- Improves Downstream Processes: Clean tablets are necessary for effective film coating, printing, and packaging.
- Enhances Product Appearance: It provides a clean, professional finish for the final dosage form.
Simultaneously, a powerful dust collection system works to keep the manufacturing area clean. It effectively absorbs airborne dust generated within the pressing chamber. This function is vital for protecting operators from inhaling fine particulate matter. A clean environment also reduces the risk of machine wear and simplifies maintenance procedures.
Expert Insight: A fully enclosed design, often featuring a stainless steel shell, complements the dust collection system. This combination contains airborne particles, prevents external contaminants from entering the pressing zone, and makes the entire machine easier to clean and validate according to GMP standards.
Modern rotary tablet presses integrate these two functions into a single, cohesive unit. The system automatically channels tablets through a dedusting path while simultaneously vacuuming excess powder from the turret area. This seamless integration minimizes manual intervention, improves operational efficiency, and upholds the highest standards of product quality and workplace safety.
Ensuring GMP Compliance and Safety
Adherence to Good Manufacturing Practices (GMP) and operator safety are non-negotiable in pharmaceutical production. A rotary tablet press must incorporate specific design features that uphold these standards. These elements protect the product from contamination and the operator from harm.
Stainless Steel Construction and Design
The choice of material for a tablet press is fundamental to GMP compliance. A fully enclosed stainless steel shell is the industry standard. The exterior and internal work surfaces should be made of stainless steel to maintain surface gloss and prevent cross-contamination. Global regulatory bodies like the FDA, EU GMP, and WHO recognize stainless steel for its biocompatibility and its ability to support validated cleaning processes.
Regulatory agencies have issued warning letters citing corrosion on equipment surfaces that contact the product. Materials like stainless steel resist corrosion, which prevents microbial adhesion and simplifies cleaning, ensuring product purity.
Transparent Protective Covers
Modern tablet presses feature transparent protective covers. These covers serve a dual purpose. They provide a clear, unobstructed view of the machine's operation, allowing operators to monitor the tableting process in real time. Simultaneously, they act as a physical barrier, containing dust within the pressing chamber and preventing accidental contact with moving parts. For practical maintenance, these covers can be fully opened, giving technicians easy access for cleaning and servicing.
Emergency Stop Devices and Safety Features
Robust safety mechanisms are essential to protect both operators and the equipment. An emergency stop device is a mandatory feature that can halt all machine functions instantly. Beyond this, advanced presses integrate safety interlock switches that meet international standards like ISO 14119. These systems ensure the machine cannot start or operate unless all protective doors and covers are securely closed.
Key safety features often include:
- Safety interlock switches that prevent machine operation when gates are open.
- Emergency stop buttons with a direct open-circuit function for immediate shutdown.
- A safety lock mechanism that secures the machine during maintenance.
These integrated features create a secure operating environment and comply with stringent certifications like CE and UL.
Noise Level During Operation
The noise level of a rotary tablet press is a critical factor for both operator safety and equipment health. High operational noise can create a hazardous work environment and may indicate underlying mechanical issues. Manufacturers must evaluate a machine's noise output, typically measured in decibels (dBA), to ensure it complies with workplace safety standards.
Regulatory bodies like the Occupational Safety and Health Administration (OSHA) set strict limits on noise exposure. Prolonged exposure to levels above 85 dBA can cause permanent hearing damage. A quieter machine protects operators and improves communication on the production floor, reducing the risk of accidents. It creates a more comfortable and less stressful work environment, which can lead to higher productivity and operator focus.
Excessive noise often signals mechanical problems. It can point to issues such as:
- Worn-out gears or bearings
- Improper lubrication
- Vibrations from misalignment
Key Insight: A well-designed machine incorporates features to minimize noise. For example, a sealed transmission system enclosed in an oil tank at the bottom of the machine body acts as an independent, lubricated unit. This design significantly reduces noise and wear by ensuring the transmission shaft operates smoothly in an oil bath.
Ultimately, a low noise level is a hallmark of superior engineering and quality construction. It demonstrates that the manufacturer has prioritized not only performance but also the long-term well-being of operators and the durability of the machine itself. Choosing a quieter press is an investment in a safer, more efficient, and more sustainable production operation.
Focusing on Operational Efficiency
Operational efficiency goes beyond production speed. It involves minimizing downtime for changeovers, cleaning, and maintenance. A well-designed rotary tablet press incorporates features that streamline these essential tasks, maximizing the machine's productive uptime.
Ease of Tooling Changeover
Tooling changeover is the process of swapping punches and dies between production runs. A machine designed for quick changeovers significantly reduces costly downtime. Features like an interchangeable turret allow operators to prepare a new toolset offline. This preparation enables a swift and simple swap, getting the press back into production much faster. Efficient changeover capabilities directly boost a facility's overall productivity and manufacturing flexibility.
Design for Quick Cleaning and Maintenance
A machine's design directly impacts the time required for cleaning and maintenance. Modern presses feature smooth, accessible surfaces and minimal dead spots to simplify cleaning procedures. Advanced systems like automated Clean-In-Place (CIP) offer major advantages over manual methods.
- Reduced Errors: CIP systems perform a consistent cleaning cycle every time, minimizing human error.
- Lower Maintenance: Automation removes the need for frequent disassembly, which cuts down on labor costs and equipment wear.
- Increased Production: These systems can reduce cleaning time by up to 90%, allowing for more production hours.
Pro Tip: Proper gasket maintenance is also crucial for hygiene. Worn gaskets can create hard-to-clean pockets where bacteria may grow. Using torque limiters and high-quality gasket materials helps maintain a sanitary production environment.
Sealed Transmission System
A sealed transmission system is a key feature for reliability and cleanliness. This system is typically enclosed in an oil tank at the bottom of the machine. It operates as an independent unit, which prevents oil from contaminating the powder in the pressing area. The transmission shaft runs in a continuous oil bath. This design provides constant lubrication, reducing operational noise and component wear. A sealed system lowers maintenance needs and contributes to a cleaner, GMP-compliant operation.
Assessing Supplier and Long-Term Costs
A smart investment in a rotary tablet press looks beyond the initial price tag. Buyers must evaluate the supplier's reliability and the full lifetime cost of the machine to ensure long-term value and operational success.
Supplier Reputation and After-Sales Support
A supplier's reputation provides insight into the quality of their products and services. Businesses should research potential suppliers by reading customer reviews, examining case studies, and verifying their standing in the industry. Strong after-sales support is equally important. A reliable supplier offers responsive technical assistance, troubleshooting, and comprehensive service agreements. This partnership ensures that expert help is available to resolve any operational issues quickly, minimizing production delays.
Total Cost of Ownership (TCO)
The Total Cost of Ownership (TCO) offers a complete financial picture of the investment. It includes all expenses incurred throughout the machine's entire lifecycle. Calculating the TCO helps a company make a more informed financial decision.
Key Insight: A machine with a lower initial price may have a higher TCO due to frequent maintenance, high energy use, or expensive parts. A comprehensive evaluation prevents unexpected long-term costs.
Key factors contributing to TCO include:
- Initial purchase price
- Installation and commissioning fees
- Ongoing energy consumption
- Routine maintenance and repair costs
- Operator training expenses
Availability of Spare Parts
Downtime is one of the biggest hidden costs in manufacturing. The ready availability of spare parts is crucial for keeping a tablet press running. A production line can sit idle for days or weeks while waiting for a critical component to arrive. Buyers should confirm that the supplier maintains a robust inventory of essential spare parts. Suppliers who offer parts compatible with other common machine models, such as the ZP series, provide an added layer of flexibility and security for maintenance operations.
Comprehensive Operator Training and Documentation
Effective operator training is essential for unlocking a machine's full potential and ensuring consistent production. A reputable supplier provides a comprehensive training program that empowers operators with deep technical knowledge. This training should extend beyond basic operations.
A thorough program covers critical areas, including:
- Machine Components: Understanding the function of the cam track system, pressure rollers, and drive system.
- Troubleshooting: Identifying factors that affect tablet weight, such as powder variation or tooling condition.
- Press Adjustments: Learning how to adjust feeder speed, scraper blades, and fill cams to overcome production issues.
- Problem-Solving: Using tools like a strobe light to observe powder flow and make precise, incremental adjustments.
The supplier must also provide clear documentation, including operational manuals, parts lists, and material certificates. This information is invaluable for daily operation and long-term maintenance.
Factory and Site Acceptance Tests (FAT/SAT)
Factory and Site Acceptance Tests (FAT/SAT) are crucial quality gates in the procurement process. The FAT occurs at the supplier’s facility before shipment. It allows the buyer to verify that the machine meets all contractual specifications.
FAT Verification Checklist: A typical FAT protocol confirms key details like the machine's make and model, materials of construction (MOC), control panel functions, and the proper operation of all safety features, including emergency stops.
Any deviations are documented and corrected before the machine leaves the factory. The Site Acceptance Test (SAT) is performed after installation at the buyer's facility. It re-validates machine performance in its actual operating environment, ensuring a seamless transition into production.
Installation and Commissioning Support
A supplier's role should not end at delivery. Professional installation and commissioning support ensures the machine is set up correctly and performs optimally from day one. This support package typically includes mechanical and electrical installation, equipment startup, and performance tuning. The supplier’s technicians configure all parameters and conduct test runs to optimize the machine for the buyer's specific formulations. This hands-on support, combined with technical guidance for operators, guarantees a smooth and efficient start to production.
Buyers should use these factors to create a detailed checklist for comparing different models. This process helps prioritize non-negotiable requirements for production, quality, and long-term support. Choosing the right rotary tablet press machine represents a strategic investment in a company's future efficiency and product quality.
Final Step: Always request live demonstrations and detailed quotes from suppliers before finalizing your decision. This ensures the machine performs as expected with your specific formulations.
FAQ
What makes a forced feeding system important?
A forced feeding system ensures uniform powder delivery into the die cavities. This guarantees consistent tablet weight and active ingredient content. It is especially crucial for high-speed production and for powders that do not flow easily, improving overall product quality.
Why is a sealed transmission system beneficial?
A sealed transmission system isolates lubrication oil from the pressing area. This design prevents product contamination. It also reduces operational noise and component wear by keeping the transmission shaft constantly lubricated, which lowers maintenance needs and supports GMP compliance.
Can one machine produce different tablet sizes?
Yes. Modern presses often feature interchangeable turrets. This capability allows manufacturers to use different tooling types, like B and D, on a single machine. This flexibility enables the production of various tablet sizes and shapes to meet diverse product requirements.
How do transparent covers improve safety and compliance?
Transparent covers provide a clear view of the operation while containing dust. They also prevent accidental contact with moving parts. The ability to open them fully simplifies cleaning and maintenance, helping operators meet strict GMP standards.
See Also
Unveiling Top 5 Applications for Single-Stage Rotary Vane Vacuum Pumps
Essential Guide: Selecting the Ideal Tablet Sampling Machine for Your Lab
Key Capsule Checkweighers for Buyers to Evaluate in the Year 2025
Innovative Folder Gluer Features Shaping 2025 Carton Production Lines
Three Essential Industrial Checkweighers for Every Modern Food Processing Plant