Choosing Between Automated and Manual Broken Tablet Detection
Pharmaceutical manufacturers rely on the broken tablet detector to maintain product integrity and meet regulatory requirements. Automated systems use high-resolution cameras and AI-based vision software to provide objective inspections, reduce human error, and support stringent quality standards.
| Benefit | Automated Systems | Manual Inspection |
|---|---|---|
| Consistency in inspections | Yes | No |
| Reduction of human error | Yes | No |
| Support for stringent quality standards | Yes | Limited |
| Compliance with regulatory requirements | Yes | Limited |
Manual Broken Tablet Detector Systems

How Manual Detection Works
Operators visually inspect tablets as they move along the production line. They use trays, magnifying glasses, or simple light sources to identify broken or chipped tablets. Staff members remove defective units by hand and record findings in logbooks or spreadsheets. The process relies on the experience and attention of trained personnel.
Advantages of Manual Detection
Manual detection offers flexibility for small-scale pharmaceutical operations. Teams can adapt inspection methods to unique tablet shapes or formulations. The approach requires minimal investment in equipment, making it accessible for startups or facilities with limited budgets. Operators can quickly respond to unexpected issues or changes in production batches.
Tip: Manual inspection suits environments with low production volumes or specialized tablet designs.
Limitations of Manual Detection
Manual broken tablet detector systems present several challenges in pharmaceutical production. Error rates average between 3% and 5%, significantly higher than automated alternatives. Fatigue increases mistakes, especially during long shifts. Distractions and interruptions contribute to miscounts, while small tablet sizes and visual similarities between products further complicate detection. Common limitations include:
- Lack of stringent oversight, which leads to missed defects.
- Dependence on trained personnel, creating risks if staff are unavailable.
- Lower speed, precision, and consistency compared to automated systems.
These factors can impact product quality and regulatory compliance, especially in high-volume environments.
Automated Broken Tablet Detector Systems

How Automated Detection Works
Automated broken tablet detector systems use advanced vision technology and precision sensors to inspect tablets as they move along the production line. High-resolution cameras capture images from multiple angles, while AI-trained algorithms analyze these images for cracks, chips, or other defects. The system can also weigh tablets and check for metal contamination. Operators receive real-time feedback, and the system automatically removes defective tablets using mechanical arms or pneumatic devices.
Types of Automated Systems
Manufacturers offer several types of automated broken tablet detector systems, each using different detection methods:
| Detection Method | Description |
|---|---|
| Visual Inspection | High-resolution cameras and image-processing software identify visible defects. |
| Weight Detection | Precision weighing sensors detect deviations in tablet weight. |
| Metal Detection | Electromagnetic fields identify metal contamination. |
| Leak Detection | Vacuum decay and helium leak testing ensure the integrity of sealed tablet strips. |
| Rejection Mechanisms | Pneumatic systems and mechanical arms remove defective tablets from the line. |
Benefits of Automation
Automated systems deliver significant improvements in accuracy and speed compared to manual inspection. They minimize human error and maximize yield by integrating hardware precision with intelligent software. These systems generate detailed reports on counts, batch details, and quality checks, supporting compliance and process optimization. Operators benefit from user-friendly interfaces and electronic batch records, which streamline audits and documentation.
Note: Automated broken tablet detector systems can process up to 600 containers per minute, maintaining high safety and inspection standards.
Challenges and Considerations
Pharmaceutical companies face several challenges when implementing automated systems:
| Challenge Type | Description |
|---|---|
| High Technical Complexity | Significant R&D costs for optics, vision algorithms, and mechanical handling. |
| Algorithm Robustness | Systems must adapt to various tablet colors, shapes, and coatings. |
| Validation Costs | High costs and time required for regulatory validation. |
| Long Decision Cycles | Extended timelines for adoption can affect return on investment. |
| Price Sensitivity | Short-term costs may concern some markets despite long-term benefits. |
AI-Powered Broken Tablet Detector Solutions
How AI Enhances Detection Accuracy
Artificial intelligence transforms the broken tablet detector by leveraging advanced algorithms and computer vision. AI models process real-time data, which enables rapid identification of defects that human inspectors might miss. The CBS-YOLOv8 model, for example, uses coordinate attention and BiFPN to extract and fuse features more effectively. This approach increases both detection speed and accuracy. Key enhancements include:
- Advanced algorithms improve the identification of subtle cracks and chips.
- Computer vision systems analyze thousands of tablets per hour.
- Real-time processing reduces the risk of defective products reaching the market.
Key Advantages of AI-Powered Systems
AI-powered systems deliver measurable benefits in pharmaceutical inspection. They maintain high throughput and accuracy, even during extended production runs. These systems eliminate human fatigue, ensuring consistent results. Inspection logs and timestamps provide traceability, which supports regulatory compliance and internal audits. Cost savings arise from fewer product recalls and reduced manual labor.
Tip: AI-powered solutions help companies meet the FDA’s Current Good Manufacturing Practices (cGMP) by enhancing transparency and auditability.
Implementation Considerations
Pharmaceutical companies must evaluate several factors before adopting AI-powered broken tablet detector solutions.
| Advantages of AI in Pharmaceutical Inspection | Challenges and Considerations in AI Implementation |
|---|---|
| Speed: Inspects thousands of units per hour | High initial costs |
| Accuracy: Detects microscopic defects | Need for skilled personnel |
| Consistency: Eliminates human fatigue | Integration with existing machinery |
| Traceability: Generates inspection logs | Continuous training and data updates required |
| Cost Savings: Reduces product recalls | Choosing the right vendor |
Companies must also ensure that AI systems integrate with existing production lines and receive regular updates. Skilled personnel play a critical role in maintaining and optimizing these advanced solutions.
Broken Tablet Detector Comparison Table
Accuracy and Reliability
Accuracy and reliability remain the most critical factors in pharmaceutical quality assurance. Manual inspection depends on the operator’s skill and attention, which leads to variable results. Automated and AI-powered systems deliver higher and more consistent accuracy rates. The following table compares the accuracy rates of different detection systems:
| System Type | Accuracy Rate |
|---|---|
| AI-Powered (CNN) | 88.8% |
| Previous Work 1 | 87.1% |
| Previous Work 2 | 85.6% |
| Previous Work 3 | 75.0% |
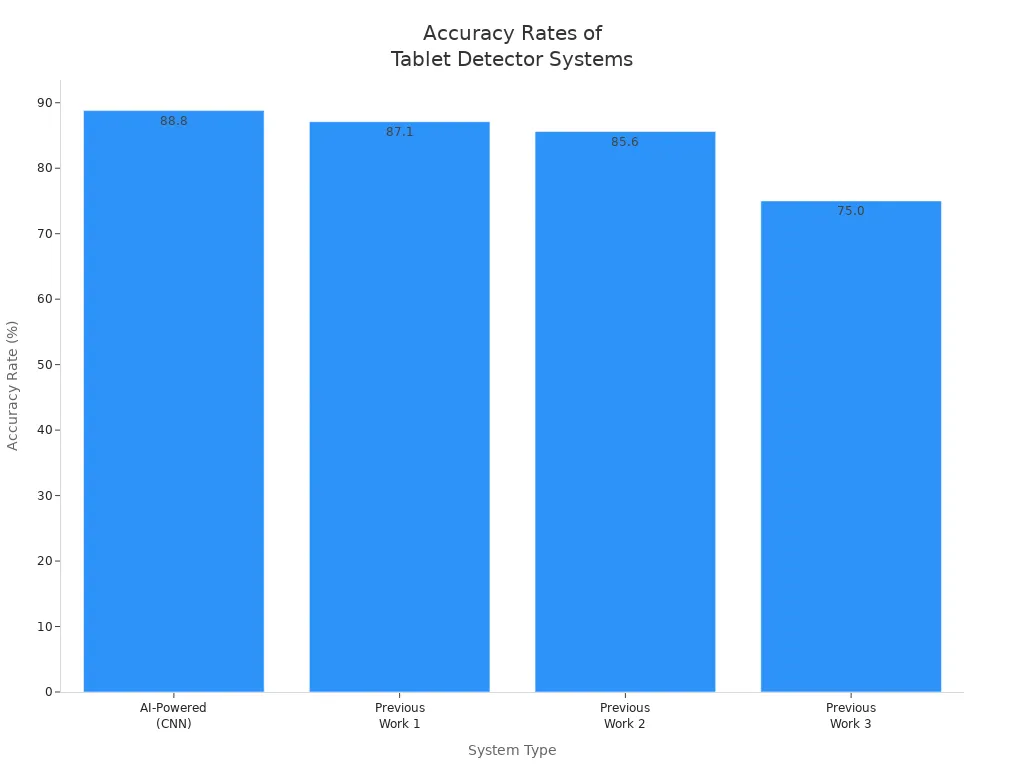
Automated systems consistently detect both macro and micro defects, while manual inspection often misses subtle cracks or chips. AI-powered solutions further enhance reliability by adapting to new tablet types and production changes without retraining operators.
Note: Automated and AI-driven systems eliminate the impact of human fatigue, ensuring uniform inspection quality across all batches.
Speed and Throughput
Speed and throughput directly affect production efficiency. Manual inspection operates at a low to moderate pace, which limits its suitability for large-scale manufacturing. Automated systems can inspect hundreds of containers per minute, maintaining high accuracy even at maximum speed. AI-powered detectors process thousands of tablets per hour, supporting real-time quality control.
- Manual inspection: Suitable for small batches or specialty products.
- Automated systems: Ideal for high-volume production lines.
- AI-powered solutions: Enable rapid scaling without sacrificing accuracy.
Cost and ROI
Cost considerations influence the choice between manual and automated systems. Manual inspection requires minimal upfront investment but incurs ongoing labor costs and higher risk of product recalls due to missed defects. Automated and AI-powered systems demand higher initial investment but deliver long-term savings through reduced labor, fewer recalls, and improved yield.
| Parameter | Manual Inspection | Automated/AI-Powered Systems |
|---|---|---|
| Initial Investment | Low | High |
| Ongoing Labor Costs | High | Low |
| Recall Risk | Higher | Lower |
| Long-Term ROI | Limited | Significant |
Tip: Companies with high production volumes often achieve a faster return on investment by adopting automated or AI-powered broken tablet detector systems.
Ease of Use and Training
Manual inspection relies on trained personnel who must maintain focus and accuracy throughout each shift. Training new operators can be time-consuming, and results may vary between individuals. Automated systems feature user-friendly interfaces and require less operator intervention. AI-powered detectors adapt to new products with minimal retraining, reducing the learning curve for staff.
- Manual: Operator-dependent, requires ongoing training.
- Automated: Intuitive controls, less training required.
- AI-powered: Self-learning capabilities, minimal retraining.
Compliance and Documentation
Meeting regulatory standards is essential in pharmaceutical manufacturing. Manual inspection often struggles with documentation and traceability, making regulatory audits challenging. Automated systems generate digital logs and detailed reports, supporting compliance with cGMP and CFR requirements. These systems also ensure audit readiness and maintain thorough documentation.
| Aspect | Description |
|---|---|
| Compliance with cGMP | Automated systems help ensure the absence of contaminants, meeting regulatory standards. |
| Patient Safety | They prevent the release of contaminated products, safeguarding patient health. |
| Quality Control | Integral to quality control processes, ensuring product integrity and safety. |
| Audit Preparedness | Robust systems ensure readiness for regulatory audits and maintain thorough documentation. |
Regulatory agencies favor automated systems for their traceability and audit-friendly features.
Scalability
Scalability determines how well a detection system supports business growth. Manual inspection cannot keep pace with increasing production volumes and requires more staff as output rises. Automated and AI-powered systems integrate seamlessly into high-throughput environments, enabling manufacturers to scale operations efficiently. AI-driven technologies use machine learning to detect defects in real time, which accelerates inspection cycles and maintains high accuracy.
Manufacturers adopting AI-enabled counters for broken or misshapen tablets reduce reliance on human quality assurance. This shift supports operational efficiency and compliance as production demands grow.
Companies planning to expand production benefit most from scalable automated or AI-powered broken tablet detector solutions.
Choosing the Best Broken Tablet Detector for Your Needs
Factors to Consider
Selecting the right broken tablet detector requires a careful evaluation of several important factors. Pharmaceutical manufacturers must balance operational needs, regulatory requirements, and financial constraints. The following considerations help guide the decision-making process:
- Effectiveness and Reliability: Automated systems deliver consistent quality and reduce human error. Manual inspection often results in variable defect detection rates, which can compromise product integrity.
- Financial Considerations and ROI: Automated solutions require a higher initial investment. Over time, these systems lower labor costs and minimize production errors, leading to significant long-term savings. Manual inspection offers a lower setup cost but may result in higher ongoing expenses due to labor and potential recalls.
- Production Context and Adaptability: Manual inspection provides flexibility for small or medium production volumes. Automated systems excel in large-scale environments where speed and uniformity are critical.
- Regulatory Compliance: Automated and AI-powered systems generate detailed inspection logs, supporting compliance with cGMP and other regulatory standards. Manual methods may struggle to provide the same level of documentation.
- Scalability: Companies planning to expand production benefit from systems that can scale with increased demand. Automated and AI-powered detectors integrate easily into high-throughput lines.
Tip: Companies should assess current production volumes, future growth plans, and compliance needs before investing in a new detection system.
Typical Use Cases
Different pharmaceutical operations require different detection solutions. Understanding typical use cases helps manufacturers match technology to their unique requirements.
- Small-Scale or Niche Production: Manual inspection suits facilities with low output or specialized tablet designs. Operators can adapt inspection methods quickly for unique products or short runs.
- High-Volume Manufacturing: Automated systems fit large-scale production lines. These systems maintain high speed and accuracy, ensuring consistent quality across thousands of units.
- Advanced Quality Control: AI-powered systems detect defects in both tablets and capsules. They achieve up to 99.9% defect detection accuracy and process over 400 units per minute. These systems also monitor blister packs for missing or damaged units, providing comprehensive quality assurance.
- Regulatory-Driven Environments: Facilities facing strict regulatory scrutiny benefit from automated or AI-powered solutions. These systems generate digital records and support audit readiness.
| Use Case | Recommended Solution | Key Benefit |
|---|---|---|
| Small batch, custom products | Manual inspection | Flexibility, low upfront cost |
| Large-scale, high-speed lines | Automated system | Speed, consistency, scalability |
| Maximum accuracy, full traceability | AI-powered detector | Highest precision, regulatory support |
Note: The right broken tablet detector not only improves product quality but also supports business growth and regulatory compliance.
The table below highlights key differences between manual and automated systems for broken tablet detector selection:
| Feature | Manual Systems | Automated Systems |
|---|---|---|
| Error Rate | >5% | <1% |
| Efficiency | Low, not scalable | High, up to 6,000 tablets/min |
| Compliance | Manual checks | Meets FDA and EU GMP standards |
| Labor Costs | Higher | 98% lower per 10,000 units |
| ROI | Longer | 18–24 months for high volume |
Industry experts recommend these steps:
- Consult equipment suppliers for tailored advice.
- Use built-in monitoring software to improve accuracy.
- Schedule routine performance checks and onsite validation.
A company should match its broken tablet detector choice to production scale, compliance needs, and long-term ROI.
FAQ
What is the main difference between manual and automated broken tablet detection?
Manual detection relies on human inspection. Automated systems use cameras and software for consistent, high-speed quality control.
Can automated systems handle different tablet shapes and sizes?
Yes. Most automated detectors adjust settings or use AI algorithms to inspect various tablet shapes, sizes, and coatings without manual recalibration.
How do automated systems support regulatory compliance?
Automated systems generate digital inspection logs. These records help companies meet FDA and cGMP requirements during audits.
See Also
A Comprehensive Guide to Choosing Tablet Sampling Machines
Is It Worth Investing in Automated Virus Sampling Lines?
The Importance of Capsule Sorters in Eliminating Defects