How 2 5-Furandicarboxylic Acid Is Used and Dissolved in Industry

You encounter 2 5-Furandicarboxylic acid in high-volume packaging, automotive, and specialty chemical manufacturing. Plants produce nearly 1.5 million metric tons annually, with packaging consuming 46% of global demand. You must understand its dissolution in water and organic solvents for efficient processing.
| Aspect | Data / Figures |
|---|---|
| Annual Production | ~1.5 million tons |
| Packaging Use | 46% of demand |
2 5-Furandicarboxylic Acid: Definition and Industrial Significance
Chemical Properties and Structure
You work with 2 5-Furandicarboxylic acid as a renewable compound derived from biomass, featuring a furan ring with carboxyl groups at the 2 and 5 positions. This unique structure gives the molecule high reactivity, especially in catalytic oxidation and polymer synthesis. Industrial processes often use liquid-phase catalytic oxidation of 5-hydroxymethylfurfural (HMF) with a Co–Mn–Br catalyst system. You control reaction parameters like catalyst concentration, temperature, and solvent composition to optimize yield and prevent catalyst deactivation. The rigid furan ring in 2 5-Furandicarboxylic acid increases the glass transition temperature and thermal stability of resulting polymers, making them suitable for demanding applications.
Tip: The furan ring and carboxylic acid groups in 2 5-Furandicarboxylic acid enable selective oxidation under mild conditions, which is critical for large-scale synthesis and efficient polymer production.
Role in Sustainable Manufacturing
You help reduce your facility’s carbon footprint by choosing 2 5-Furandicarboxylic acid for bio-based polymer production. Unlike petroleum-based alternatives, this compound comes from renewable plant sugars, lowering fossil fuel dependence and greenhouse gas emissions. The manufacturing process uses milder reaction conditions, which decreases energy consumption and aligns with green chemistry principles.
| Environmental Benefit | FDCA (Bio-based) | Petroleum-based Alternatives |
|---|---|---|
| Raw Material Source | Renewable biomass | Non-renewable petroleum |
| Energy Consumption | Lower | Higher |
| Carbon Footprint | Reduced | Higher |
| Environmental Impact | Less pollution | Significant pollution |
| Sustainability | Supports renewable | Relies on finite resources |
You support a transition to a low-carbon economy and promote sustainable, circular manufacturing systems by adopting FDCA-based materials. These choices contribute to long-term environmental health and regulatory compliance.
Industrial Uses of 2 5-Furandicarboxylic Acid

Bio-Based Polymers and Plastics (PEF)
You see the most significant impact of 2 5-Furandicarboxylic acid in the production of bio-based polymers, especially polyethylene furanoate (PEF). PEF stands out as a renewable alternative to PET, offering you several advantages in packaging and sustainability. When you use PEF, you benefit from:
- Higher gas barrier properties for oxygen, carbon dioxide, and water vapor compared to PET.
- Suitability for packaging applications such as bottles, films, and food trays.
- A 100% recyclable, non-toxic, and bio-based polymer made by polymerizing FDCA with ethylene glycol.
- Superior thermo-chemical, mechanical, and recyclability properties over PET and PBT.
- Significant reductions in greenhouse gas emissions and non-renewable energy use when you replace PTA with FDCA in PEF production.
You can see the performance differences between PEF and PET in the table below:
| Property | PEF (from 2,5-FDCA) | PET (polyethylene terephthalate) | Comparison / Notes |
|---|---|---|---|
| Young's Modulus | ~2.0 GPa | ~1.3 GPa | PEF has significantly higher stiffness. |
| Yield Stress | Higher than PET | Lower | PEF shows higher yield stress due to motional constraints. |
| Elongation at Break | ~450% (at sufficient molecular weight) | Lower, more ductile overall | PEF can be ductile but tends to be slightly more brittle than PET. |
| Glass Transition Temp. | 75–80°C | Slightly lower | PEF has a higher Tg, indicating better thermal resistance. |
| Oxygen Permeability | Reduced by factor of ~11 | Baseline | PEF has much better oxygen barrier properties. |
| Carbon Dioxide Permeability | Reduced by factor of ~19 | Baseline | PEF shows greatly improved CO2 barrier. |
| Water Vapor Permeability | Reduced by factor of ~2.1 | Baseline | PEF also improves water vapor barrier, though less dramatically than gases. |
| Barrier in Stretched Samples | O2: 10.4× improvement; CO2: 12.7× improvement | Baseline | Barrier improvements persist after biaxial stretching, relevant for bottle applications. |
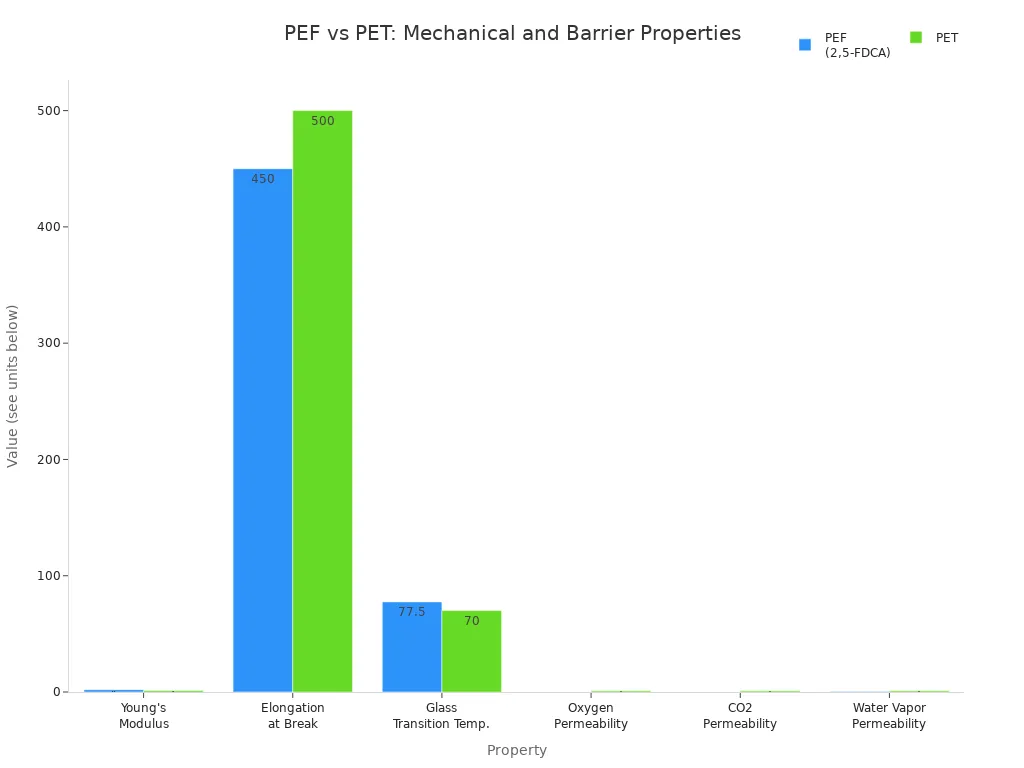
You notice that PEF exhibits six times higher oxygen barrier, three times higher carbon dioxide barrier, and double the water vapor barrier compared to PET. These properties make PEF a top choice for food and beverage packaging, where you need to extend shelf life and reduce spoilage. The demand for PEF continues to rise as more companies seek sustainable packaging solutions.
Note: Over the past decade, you have seen a clear upward trend in the demand for 2 5-Furandicarboxylic acid, especially in green polymer production and packaging. New plants and advances in catalytic oxidation methods support this growth.
Specialty Chemicals, Surfactants, and Resins
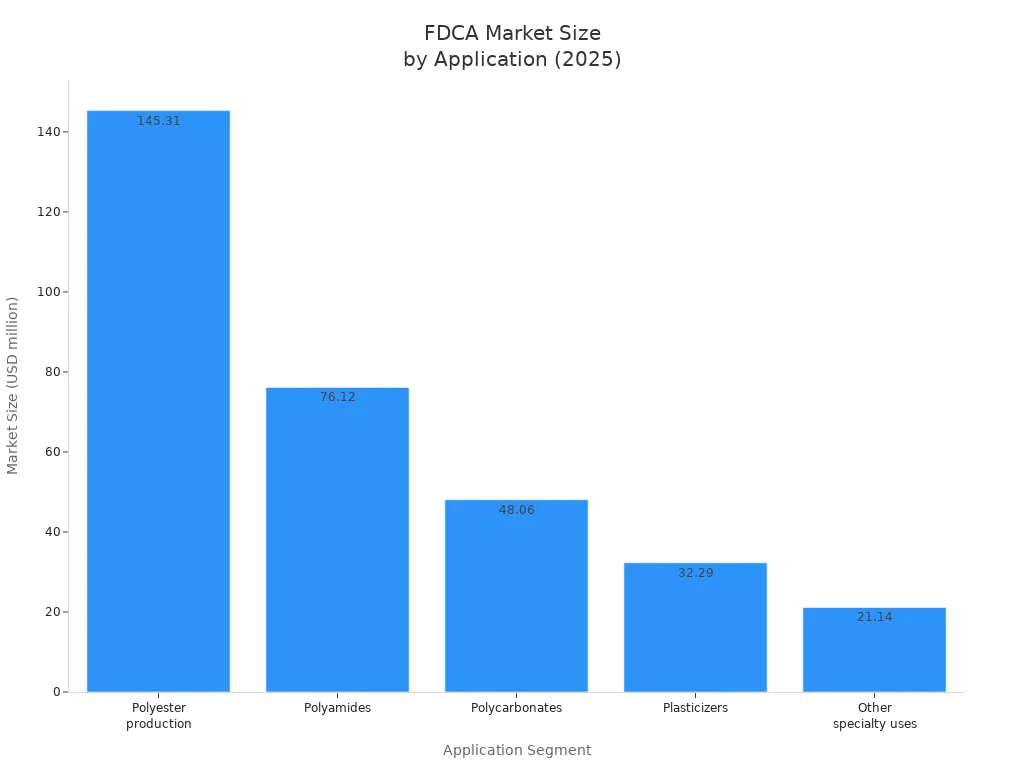
You also use 2 5-Furandicarboxylic acid as a building block for specialty chemicals and resins. This compound enables you to produce a wide range of bio-based polymers, including polyamides, polycarbonates, plasticizers, and polyester polyols. These materials serve industries such as automotive, textiles, electronics, and consumer goods.
- Polyamides made from FDCA offer you strong mechanical properties and thermal stability, making them suitable for engineering plastics and automotive parts.
- Polycarbonates and polyester polyols derived from FDCA provide you with specialty polymers for coatings, adhesives, and foams.
- Plasticizers based on FDCA help you enhance polymer flexibility in various applications.
| Application Type | Description | Market Share / Growth |
|---|---|---|
| PET (PEF) | Bio-based polyester for sustainable packaging, especially bottles | Largest market revenue share: 43.2% in 2024 |
| Polyamides | Engineering plastics, textiles, automotive parts | Fastest CAGR expected from 2025 to 2032 |
| Polycarbonates | Specialty polymers for diverse applications | Included in market segmentation |
| Plasticizers | Enhance polymer flexibility | Included in market segmentation |
| Polyester Polyols | Used in polyurethanes and resins | Included in market segmentation |
You gain several advantages by choosing FDCA-based polymers over traditional raw materials:
| Advantage Category | Description |
|---|---|
| Sustainability | FDCA is bio-based and renewable, derived from non-food biomass like corncobs and sawdust. |
| Environmental Impact | Supports carbon reduction policies and reduces reliance on petroleum-based raw materials. |
| Performance of Polymers | FDCA-based polymers show superior heat resistance, mechanical strength, and gas barrier properties. |
| Versatility in Polymer Types | You can replace terephthalic acid, isophthalic acid, and bisphenol A in polyesters, polyamides, and resins. |
| Recyclability | FDCA-based polymers are more sustainable and recyclable than traditional petroleum-based polymers. |
Tip: The chemical segment, including specialty chemicals and resins, dominates FDCA applications with a 56.7% market share in 2024. Packaging, automotive, textiles, and electronics are the main end-use sectors.
Fire Extinguisher Foams and Emerging Applications
You find that 2 5-Furandicarboxylic acid is gaining attention in new and emerging applications. Researchers and manufacturers explore its use in fire extinguisher foams, where you need environmentally friendly and effective alternatives to traditional agents. FDCA-based foams can offer you improved biodegradability and reduced toxicity, aligning with stricter environmental regulations.
You also see ongoing research into using FDCA in advanced materials, such as:
- High-performance composites for automotive and aerospace industries.
- Biodegradable plastics for single-use items.
- Specialty coatings and adhesives with enhanced durability.
You notice that the market for FDCA is expanding rapidly. Packaging remains the largest sector, but you see strong growth in textiles, automotive, consumer goods, and electronics. The global market size is projected to grow from USD 480 million in 2023 to USD 1,980 million by 2032.
Note: As you adopt FDCA-based materials, you support a shift toward sustainable, circular manufacturing systems and help meet evolving regulatory and consumer demands.
2 5-Furandicarboxylic Acid Solubility and Industrial Handling

Solubility in Water and Organic Solvents
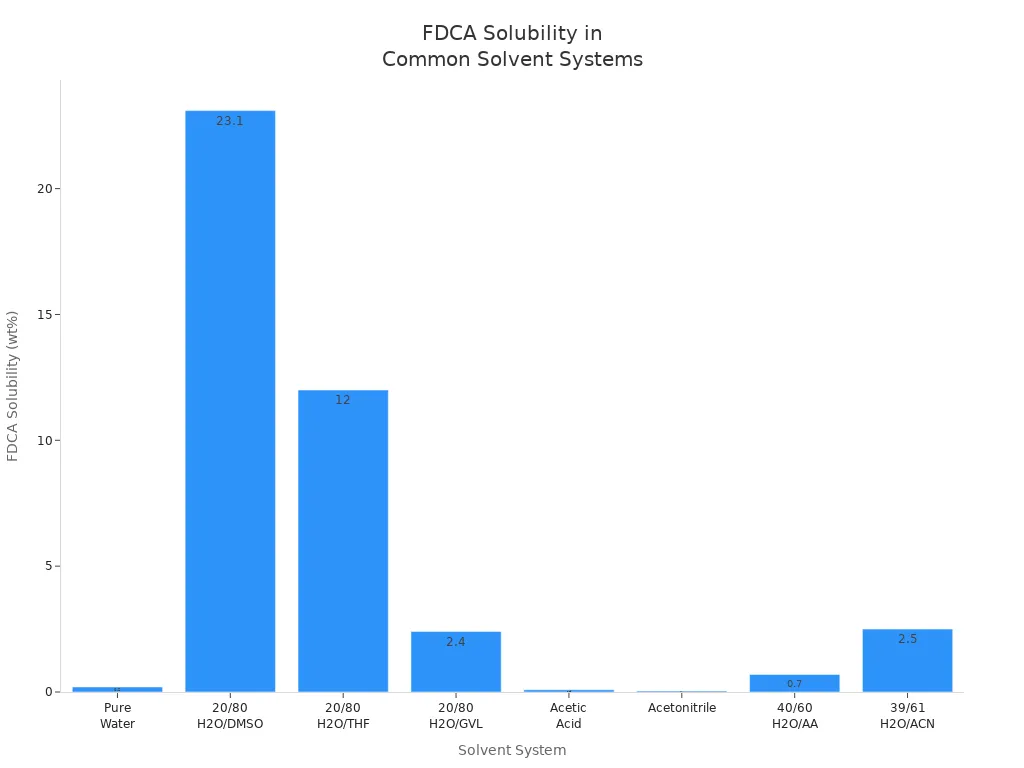
You often face challenges when dissolving 2 5-Furandicarboxylic acid in industrial settings. This compound shows low solubility in pure water, which can limit its direct use in aqueous processes. You can dramatically improve solubility by using organic solvents or solvent blends. For example, mixing water with dimethyl sulfoxide (DMSO), tetrahydrofuran (THF), or gamma-valerolactone (GVL) increases FDCA solubility by up to 190 times compared to pure water. Methanol and ethanol also provide high solubility, making them popular choices for laboratory and pilot-scale work.
| Solvent System | Temperature (K) | FDCA Solubility (wt %) | Notes on Solubility Parameter Correlation |
|---|---|---|---|
| Pure Water | 293 | ~0.2 | Low solubility; baseline for comparison |
| Pure DMSO | 293 | Higher than water | Used in blends to enhance solubility |
| 20/80 w/w H2O/DMSO | 293 | 23.1 | 190x solubility increase vs pure water |
| 20/80 w/w H2O/THF | 293 | ~12 | 60x solubility increase vs pure water |
| 20/80 w/w H2O/GVL | 303 | 2.4 | 10x increase over pure water |
| Acetic Acid (AA) | 323 | 0.09 | Lower than pure water at same T |
| Acetonitrile (ACN) | 323 | 0.04 | Lower than pure water at same T |
| 40/60 w/w H2O/AA | 323 | 0.70 | ~2x increase over pure components |
| 39/61 w/w H2O/ACN | 323 | 2.5 | Significant increase over pure components |
| Pure Methanol (MeOH) | 293 | High solubility | Among highest in pure solvents |
| Pure Ethanol (EtOH) | 293 | High solubility | Similar to MeOH |
You can see the dramatic effect of solvent choice in the chart below:
Temperature and pH also play a critical role. When you increase the temperature, FDCA solubility rises in mixed solvents like GVL/water. You can tune solvent composition and temperature to maximize dissolution, which is essential for efficient extraction and processing. In biphasic systems, pH adjustment helps you control FDCA distribution between water and organic phases, optimizing extraction yields.
Tip: You should always consider both solvent blend and temperature when designing FDCA dissolution steps. Small changes can lead to major improvements in process efficiency.
Challenges in Dissolution and Processing
You encounter several obstacles when processing 2 5-Furandicarboxylic acid at scale. Low solubility in water complicates direct handling and extraction. Biomass-derived feedstocks introduce impurities such as amino acids, which poison noble metal catalysts and reduce FDCA yield. Side products like levulinic and formic acid further disrupt catalyst activity and process control.
| Challenge | Description | Impact on Industrial FDCA Processing |
|---|---|---|
| Catalyst deactivation by impurities | Amino acids from biomass adsorb on catalysts, poisoning them | Reduces catalyst activity and FDCA yield |
| Feedstock variability and side products | Levulinic and formic acid lower catalyst performance | Lowers FDCA yield, complicates process control |
| Catalyst separation and recycling | Homogeneous catalysts are hard to separate and recycle | Increases process complexity and cost |
| Gas–liquid mass transfer limitations | Oxygen flux through gas–liquid films limits reaction rates | Reduces productivity and scalability |
| Scale-up challenges | Maintaining catalyst stability and high yields in continuous processes | Difficult to sustain performance at industrial scale |
| Catalyst stability improvements | Alloying Pd with Au improves tolerance to poisoning | Enables longer catalyst life and better sustainability |
Long reaction times often result from gas–liquid mass transfer resistance, especially when oxygen must diffuse through liquid films. You may find homogeneous catalysts effective for FDCA synthesis, but their separation and recycling add complexity. Heterogeneous catalysts such as platinum, palladium, or gold on supports offer better practicality for large-scale operations.
Note: You must carefully manage feedstock purity, catalyst selection, and reactor design to overcome these challenges and maintain high FDCA yields.
Industrial Solutions and Process Modifications
You can address solubility and processing challenges by adopting advanced technologies and process modifications. Biocatalytic oxidation of HMF to FDCA using whole-cell or enzymatic systems allows you to operate under mild conditions, improving selectivity and conversion efficiency. Enzymatic cascades with oxidases and laccases further enhance yields.
- Biocatalytic methods provide eco-friendly alternatives and potential for scale-up.
- Chemical catalytic oxidation requires high temperatures and noble metal catalysts, but you can improve economics by switching to heterogeneous catalysts and closed-loop recycling.
- Genetic manipulation of whole-cell systems and product purification remain areas for improvement.
| Process Step / Technology | Description | Industrial Equipment / Technology |
|---|---|---|
| Biomass Pretreatment & Fractionation | Conversion of waste to xylose-rich hydrolysates | Pretreatment reactors, fractionation units |
| Dehydration & Oxidation | Furfural conversion at pilot scale | Reactive distillation, continuous reactors |
| Carboxylation & Catalyst Regeneration | Suspension carboxylation with catalyst recycling | Carboxylation reactors, regeneration systems |
| Product Purification | Separation and purification for downstream use | Purification units for polyester synthesis |
| Catalyst and Solvent Recycling | Closed-loop systems for sustainability | Solvent recovery, catalyst recycling |
You can optimize flow rates, temperature, and pressure in continuous packed-bed reactors to maintain high yields and productivity. Advanced purification techniques help you obtain polymer-grade FDCA, which is crucial for high-performance applications.
Alert: You should always evaluate the environmental impact of your process. Green metrics like E-factor and Process Mass Intensity (PMI) help you measure sustainability and guide process improvements.
Practical Examples from Industry
You can learn from several successful industrial implementations of FDCA production and handling:
- Avantium’s ‘YXY’ process converts carbohydrates to methoxymethylfurfural and then oxidizes to FDCA, with pilot plants producing up to 50,000 tons/year.
- Corbion uses microbial biotransformation for large-scale FDCA production, collaborating with BASF in the Succinity consortium.
- DuPont and Archer Daniels Midland Company developed high-pressure oxidation processes for FDCA esters.
- Origin Materials and Eastman launched commercial FDCA production in 2017.
- AVA Biochem started with 30,000 tons/year FDCA production, aiming for 120,000 tons/year.
- Petrobras uses a two-step process with resin catalysts for FDCA conversion from sugars.
- BASF achieved a 95.2% FDCA yield using Pt/C catalyst under high pressure and temperature.
Industry leaders emphasize the importance of sustainable synthesis, effective purification, and continuous process development. You see that polycondensation of FDCA with ethylene glycol produces PEF, a bio-based polyester with superior mechanical and barrier properties compared to PET. FDCA’s versatility extends to food, medical, and aviation materials, highlighting its broad industrial relevance.
Callout: You should focus on process scalability, catalyst stability, and purification to ensure consistent product quality and environmental sustainability.
You see 2 5-Furandicarboxylic acid driving sustainable plastics, high-performance fabrics, and coatings. Its poor solubility and thermal instability require innovative processing, such as esterification.
Ongoing research and market growth in Asia-Pacific promise broader adoption, especially as industries seek eco-friendly, bio-based materials.
| Challenge | Solution |
|---|---|
| Low solubility | Esterification, blends |
| High costs | Tech innovation |
FAQ
What makes 2 5-Furandicarboxylic acid important for sustainable plastics?
You choose 2 5-Furandicarboxylic acid because it comes from renewable sources. It helps you create bio-based plastics with lower carbon footprints and improved barrier properties.
How do you improve the solubility of 2 5-Furandicarboxylic acid in industrial processes?
You increase solubility by using solvent blends like water with DMSO or THF. Raising the temperature also helps you dissolve it more efficiently.
Can you recycle polymers made from 2 5-Furandicarboxylic acid?
Yes, you can recycle these polymers. They offer you better recyclability than many petroleum-based plastics, supporting your circular manufacturing goals.